RICSA, 3(1),67-72. (2026)
Etoricoxib-Induced Fixed Drug Eruption
with Cutaneous and Mucosal
Involvement: A Case Report
Eritema
pigmentado fijo en brazos y mucosa oral posterior a la ingesta
de etoricoxib: reporte
de caso.
Luz M. Moyano Vidal1,2,3,a,b; Lucia M. Bolivar-Herrada4,c; Martha P. Diaz-Guevara5,b; from the INERTROP working group.
Filiación:
1. Unidad Médico Legal Contralmirante Villar,
Instituto de Medicina
Legal y Ciencias
Forenses, Tumbes, Perú.
2. Escuela Profesional de Medicina Humana,
Facultad de Ciencias
de la Salud, Universidad Nacional
de Tumbes, Tumbes,
Perú.
3. Doctora en Ciencias
de Medio Ambiente
y Sociedad, con mención en Medio Ambiente
y Salud Pública, Tumbes, Perú.
4. Establecimiento de Salud Pampa Grande, Dirección Regional de Salud
de Tumbes, Tumbes,
Perú.
5. Unidad Médico Legal
II Tumbes, Instituto de Medicina Legal
y Ciencias Forenses,
Tumbes, Perú.
a. Médica cirujana investigadora Renacyt nivel I.
b. Médica Legista
c. Medica especialista en medicina familiar
y comunitaria
Correspondencia: Luz María Moyano Vidal,
luzmariamoyano@gmail.com
ID ORCID
de los autores
Luz
María Moyano Vidal https://orcid.org/0000-0002-5878-5782 Lucia Margarita Bolivar Herrada
https://orcid.org/0009-0007-3337-1560 Martha Patricia Diaz Guevara
https://orcid.org/0009-0009-7771-6086
Authorship statement:
Writing–original draft: LMMV, LMBH, MPDG.
Writing–review & editing:
LMMV, LMBH, MPDG. Supervision: LMMV, LMBH, MPDG. Final
approval of the manuscript: all authors.
Conflict of interest
statement: The author declares
that they have no financial, personal, or institutional conflicts of interest that could influence the results or
interpretation of the study.
Funding: This study was funded with the author’s
own resources and did not receive external
funding from
public or private institutions.
Ethical Considerations: The patient
described in this case report is the corresponding author.
Informed written consent
was obtained prior to submission for the publication of clinical data and photographic material with full anonymization. The corresponding author
acknowledges the potential for reporting bias inherent in first-person case descriptions and has ensured
objective documentation of clinical findings based on contemporaneous medical records. No institutional ethics
committee approval was required, as this constitutes a particular case report without experimental intervention. The present
study was conducted
in accordance with the Declaration of Helsinki and applicable national regulations.
Artificial Intelligence Use Declaration: During the preparation of this manuscript, Luz M. Moyano (LMMV) used Claude AI Pro (Anthropic), Perplexity
AI Pro, Open Evidence, Medsearch, and Scopus AI for the following purposes: integrating clinical history
data, reducing word redundancy, improving spelling and grammar, enhancing paragraph clarity, conducting
real and updated scientific evidence searches, structuring the discussion section, and confirming
references. After using these tools/technologies, the authors reviewed and edited the content as needed and assumed
full responsibility for the last version of the publication.
Recibido: 20-03-2026.
Aceptado: 30-03-2026.
Publicado: 31-03-2026.
Esta obra
está publicada bajo la licencia
CC BY 4.0
DOI: http://doi.org/10.57188/ricsa.2026.010
ABSTRACT
Introduction: Fixed drug eruption
(FDE) is a delayed hypersensitivity reaction characterized by erythematous- violaceous plaques recurring at identical
anatomical sites upon re-exposure to the causative drug, with persistent residual hyperpigmentation. Etoricoxib, a widely used COX-2 inhibitor, is an increasingly recognized trigger. Case
Report: A 49-year-old woman developed hyperpigmented plaques on the arms
and oral mucosa eight hours after a
single dose of etoricoxib 120 mg for acute low back pain. A clinically
identical episode had occurred two
years earlier at the same sites, initially misdiagnosed as herpetic infection.
Diagnosis was established by temporal
chronology, exact anatomical recurrence, and a Naranjo score of 10 (definite
causal relationship). Management comprised drug discontinuation, oral antihistamine, and topical corticosteroid, with favorable
outpatient evolution. Conclusion: This case illustrates
etoricoxib-induced FDE with cutaneous and mucosal
involvement and exact site recurrence. Early recognition, definitive
discontinuation, and pharmacovigilance
reporting are clinical imperatives to prevent recurrence and ensure safe
alternatives.
Keywords: Fixed drug eruption; Etoricoxib;
Cyclooxygenase-2 Inhibitors; Drug Hypersensitivity; Pharmacovigilance. (Source: MeSH-NLM
RESUMEN
Introducción: La erupcio´n fija medicamentosa
(EFM) es una reaccio´n de hipersensibilidad retardada caracterizada por placas eritematoviola´ceas recurrentes
en los mismos sitios anato´micos tras la reexposicio´n al fa´rmaco causal, dejando
hiperpigmentacio´n residual. Etoricoxib, inhibidor selectivo de COX-2, es un desencadenante. Reporte del caso: Mujer de 49 an˜os desarrollo´ placas hiperpigmentadas en brazos y mucosa oral
ocho horas despue´s de etoricoxib 120 mg para lumbalgia aguda. Un episodio
ide´ntico ocurrio´ dos an˜os antes en las mismas localizaciones, interpretado como infeccio´n herpe´tica. El diagno´stico de EFM se establecio´
por cronologí´a
temporal, recurrencia exacta y puntuacio´n de 10/13 del Naranjo
Probability Scale. El manejo incluyo´
suspensio´n del fa´rmaco,
antihistamí´nico oral y corticoide to´pico, con evolucio´n favorable. Conclusión: Este caso ilustra un
EFM por etoricoxib con compromiso cuta´neo y mucoso, de recurrencia exacta. El
reconocimiento temprano, suspensio´n
definitiva y reporte a farmacovigilancia son imperativos clí´nicos para
prevenir recurrencias y garantizar
alternativas seguras.
Palabras clave: Erupcio´n fija medicamentosa; Etoricoxib; Inhibidores de ciclooxigenasa-2; Hipersensibilidad a medicamentos; Farmacovigilancia.
INTRODUCTION
Etoricoxib
is a selective cyclooxygenase-2 (COX-2) inhibitor
widely prescribed for musculoskeletal pain
and chronic inflammatory conditions, often perceived
as a safer alternative to non-selective NSAIDs
in terms of gastrointestinal tolerability.(1,2)
However, multiple adverse cutaneous
reactions have been described in association
with its use, ranging from localized eruptions
to severe toxic epidermal necrolysis, prompting
reassessment of its dermatological safety
profile.(1,3-5)
Among
these reactions, fixed drug eruption (FDE) is
characterized by well-demarcated erythematous-violaceous
macules or plaques that invariably recur at the same anatomical sites upon re-exposure
to the causative drug and leave persistent
residual hyperpigmentation.(1–4) In clinical
practice, NSAIDs and particularly selective COX-2
inhibitors are recognized as frequent triggers
of this delayed hypersensitivity pattern, with
a growing number of reports specifically linking
etoricoxib to both cutaneous and mucosal FDE.(1-4,7)
Several
recent cases have documented etoricoxib- induced
FDE with localized and generalized distribution, including bullous lesions and
oromucosal
involvement, thereby broadening the clinical
spectrum associated with this agent and highlighting
the importance of including it in the differential diagnosis
of recurrent hyperpigmented lesions and probable drug-induced oral erosions.(1-4) Cross-reactivity with other NSAIDs, varying clinical severity, and, in the
most extreme circumstances,
life-threatening cutaneous toxicity have
also been reported.(2,3,5)
Current
drug allergy guidelines emphasize that early
recognition of these patterns, timely withdrawal
of the suspected drug, and identification
of safe alternatives are fundamental to
preventing recurrences and reducing the risk of serious complications.(6) In this context,
case reports including those from
Latin American primary care settings
where etoricoxib is widely prescribed
remain a key source for characterizing the
clinical presentation, latency, and natural course
of etoricoxib-associated cutaneous reactions.(1-4,7)
This report describes two documented
episodes of etoricoxib-induced FDE with
cutaneous and mucosal involvement, diagnosed
in the primary care setting after an initial
misdiagnosis, with the aim of expanding recognition
of this entity at the first level of care.
CASE PRESENTATION
A
49-year-old woman and university professor, with
a history of myomectomy (June 2015) and regular
use of tranexamic acid for metrorrhagia secondary to uterine myomatosis, presented on 12 March
2026 with acute onset of hyperpigmented plaques
on the upper limbs and oral mucosa, appearing
eight hours after a single dose of etoricoxib
120 mg for acute low back pain, administered
concomitantly with orphenadrine 100 mg. Relevant
past medical history included an adverse
reaction to epidural anesthesia and tachycardia following
a third dose of aspirin
75 mg in 2021. Her mother had a known hypersensitivity reaction to tramadol.
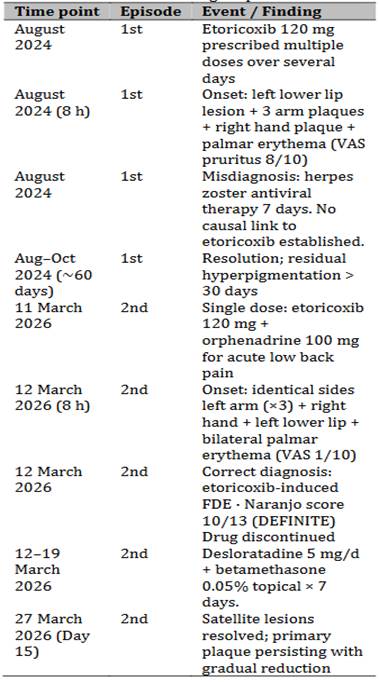
First
episode (August 2024). The patient reported a
clinically identical episode in August 2024 following
several days of etoricoxib 120 mg. Lesions
appeared on the oral mucosa (left lower lip),
initially diagnosed as herpes zoster, and treated
with antiviral therapy for seven days. Simultaneously,
three hyperpigmented plaques appeared
on the distal third of the left arm, one at the anatomical snuffbox of the right hand, and
intense
bilateral palmar erythema in the hypothenar
regions (VAS pruritus: 8/10). Lesions resolved
over 30 – 60 days with residual hyperpigmentation
lasting more than one month. No
causal link with etoricoxib was established at
that time.
Second
episode and current presentation (March 2026).
Eight hours after etoricoxib 120 mg on 11 March 2026,
the patient identified reappearance of lesions at anatomically identical sites.
Physical examination revealed three
hyperpigmented plaques on the distal
third of the lateral left arm: primary
lesion 1.0 × 1.3 cm, dark violaceous, well- demarcated, smooth surface; two satellite lesions
1.0 × 1.2 cm (medial) and 1.0 × 1.5 cm (lateral), less intense
pigmentation, diffuse borders. A 1.0 × 1.2 cm
plaque was observed at the right anatomical snuffbox. Mucosal
involvement of the left lower lip, bilateral palmar thenar erythema with
local burning, no genital or plantar lesions.
Pruritus mild (VAS 1/10) notably lower than the prior
episode. Nikolsky sign negative. (see
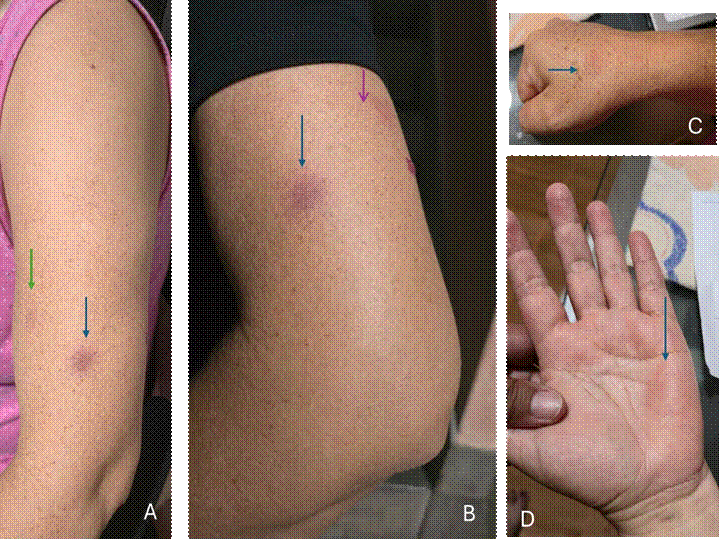
Figure 01)
Figure 1. Etoricoxib-induced fixed drug eruption
AB, left arm: primary lesion
1.0 × 1.3 cm, dark violaceous, well- demarcated,
smooth surface (black arrow); two satellite
lesions A, 1.0 × 1.2 cm (green arrow, medial) and B, 1.0 ×
1.5 cm (lateral, purple arrow), less intense pigmentation, diffuse borders. C, 1.0 × 1.2 cm plaque
was observed at the right anatomical snuffbox.
Mucosal involvement of the left lower lip. D, bilateral palmar thenar erythema
with local burning.
Vital
signs: BP 110/65 mmHg, HR 75 bpm, RR 19 rpm, axillary
temperature 36.5 °C, SpO₂ 99%. Alert, oriented, hemodynamically stable, no lymphadenopathy, no hepatosplenomegaly.
Table 1. Naranjo Algorithm
Adverse Drug Reaction
Probability Scale in the case report.
|
Questions yes no Score
|
|
Previous reports of this reaction
Reaction after suspected
|
+1
+2
|
|
|
drug:
Improved on withdrawal:
|
+1
|
|
|
Recurred on re- administration Alternative causes:
|
+2
|
+1
|
|
Reaction with placebo:
|
|
+1
|
|
Drug at toxic level:
|
|
0*
|
|
More severe at higher dose
Similar reaction to same
|
+1
|
0*
|
|
drug before
Confirmed by objective
|
+1
|
|
evidence Total score 10
 DEFINITE causal relationship (≥9 =
definite); Naranjo et al., (Clin
Pharmacol Ther 1981). Total Score ≥9: Definitive; Total Score 5 to 8. Probable; Total Score
1 to
4 Possible; Total Score
≤0. Doubtful (10)
DEFINITE causal relationship (≥9 =
definite); Naranjo et al., (Clin
Pharmacol Ther 1981). Total Score ≥9: Definitive; Total Score 5 to 8. Probable; Total Score
1 to
4 Possible; Total Score
≤0. Doubtful (10)
Differential diagnosis: Contact dermatitis excluded
(no relevant contactant exposure). Recurrent
herpetic infection ruled out (morphology,
distribution, and absence of vesicles inconsistent). Stevens-Johnson syndrome excluded (limited
extent, no epidermal
detachment, negative Nikolsky sign). Orphenadrine-induced FDE excluded: orphenadrine is not a recognized
FDE trigger, and exact lesion
recurrence across both episodes in
which etoricoxib was the common denominator
firmly attributes causality to etoricoxib.
No skin biopsy, patch testing, or oral provocation
challenge was performed; the clinical diagnosis
was conclusive, and re-exposure would represent
unnecessary risk.(see table 01).
Management. Immediate and permanent discontinuation of etoricoxib. Ambulatory regimen: desloratadine 5 mg orally once
daily × 7 days; betamethasone 0.05%
topical cream twice daily × 7 days.
No
hospitalization required. Patients
were
instructed to avoid all selective COX-2 inhibitors. Weekly
outpatient follow-up scheduled. Follow-up at 15 days. Satellite lesions resolved with progressive attenuation of hyperpigmentation. Primary left arm plaque persists with gradual reduction. For
reference, the first episode (2024)
required approximately 60 days for
complete resolution. Patient adherent, with
adequate understanding of permanent avoidance
of the causative agent.(see table 2).
Table 2. Clinical timeline of two episodes of etoricoxib-induced fixed drug eruption.
gradual reduction
The
present case is consistent with an expanding
body of literature documenting etoricoxib-induced FDE in high-impact dermatology, allergology, and oral surgery journals, as well as in Latin
American clinical
publications.(1-5,7-10) Across these reports,
the clinical presentation is relatively uniform:
well-demarcated erythematous- violaceous macules or plaques appearing hours to
days after
drug administration, exact recurrence at identical
anatomical sites upon re-exposure, and resolution
with persistent post-inflammatory hyperpigmentation.(1-4,9)
(see Table 3) In our patient, the
latency of approximately eight hours after
a single 120 mg dose, together with exact recurrence of hyperpigmented plaques
on the arms and oral mucosa at anatomically identical locations to the 2024 episode,
reproduces this classical FDE pattern
precisely.(1-3)
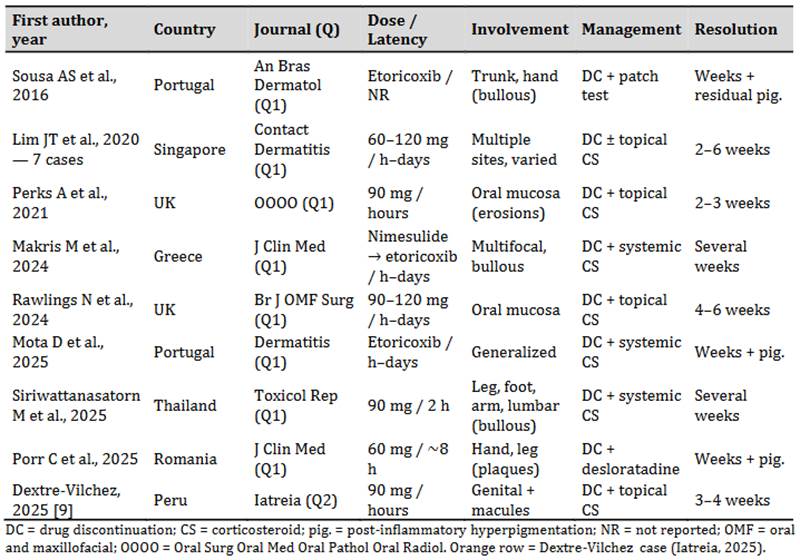
The
cases reported by Porr et al. and Makris et al. provide the closest
clinical parallels.(1,2) Porr et al. describe a woman who developed well-demarcated plaques eight hours after a single 60 mg
dose of etoricoxib, with confirmation
by oral provocation challenge.(1)
Makris et al. report cross-reactivity between
nimesulide and etoricoxib, with bullous lesions, underscoring that sensitization may extend across chemically related or even
unrelated NSAIDs.(2) Relevant to the
present report is the Peruvian case
documented by Dextre-Vilchez (2025),
which describes etoricoxib-induced FDE with
genital and mucosal involvement confirming that
this adverse reaction is recognized in our region
and that local pharmacovigilance data are beginning
to emerge.(9)
The oral mucosal involvement in this case has been documented by Rawlings et al. and Perks et al., who
report
etoricoxib-induced FDE presenting as oral erosions at high risk of misdiagnosis as herpetic or idiopathic
ulcerative conditions.(3,4) The initial management
of our patient as a herpetic infection later corrected when the second episode disclosed the causative pattern reflects precisely
this diagnostic pitfall, and
underscores the value of detailed
drug history review in recurrent oral lesions.(3,4)
Regarding
the concomitant use of orphenadrine 100
mg during the second episode: orphenadrine, a
centrally acting skeletal muscle relaxant with
anticholinergic properties, is not recognized as an FDE trigger in published literature. The occurrence of clinically identical lesions at the
same sites during the first episode
in which orphenadrine was not documented
firmly attributes causal responsibility
to etoricoxib rather than to the concomitant agent.
Table 3. Published cases of etoricoxib-induced fixed drug eruption (2016–2025)
The
Naranjo probability scale yielded a score of
10/13, classifying the causal relationship as definite (score ≥9).(11) This systematic assessment supports the diagnostic
conclusion even in the absence of
skin biopsy, epicutaneous patch
testing, or oral drug provocation challenge procedures
that, while used in confirmatory series,(1,2,8,10)
carry procedural risk and are generally
not required when clinical diagnosis is unambiguous.(6)
From
a pharmacovigilance perspective, most published etoricoxib-induced FDE cases arise from specialized services, suggesting
significant underreporting from primary care settings.(3,6–9) The present report originates in a general outpatient setting and follows the
sequence described by Makris et al.
and others: initial misdiagnosis of
the first episode, followed by pattern
recognition only when a second episode occurred.(2,6)
In Peru, adverse drug reactions can be reported
to DIGEMID (Direccio´n General de
Medicamentos, Insumos y Drogas: www.digemid.minsa.gob.pe). Strengthening mandatory adverse drug reaction reporting, particularly
for widely prescribed COX-2 inhibitors,
is essential to generate regional epidemiological data and improve drug safety monitoring at the primary care level.
This report describes a particular case without skin biopsy, patch testing, or oral provocation
challenge. As a first-person
case report, the potential for reporting bias mitigated by objective clinical
documentation must be acknowledged. Systematic photographic
documentation of the first episode was
not available, limiting longitudinal visual assessment. These limitations are notwithstanding, the definite Naranjo score (10-13), exact anatomical recurrence across two
documented episodes, and concordance
with the international published
series support the diagnosis robustly.
This report documents two consecutive episodes
of etoricoxib-induced fixed
drug eruption with cutaneous
involvement of the upper limbs and mucosal
involvement of the lip, characterized by short
latency (~8 hours), exact anatomical recurrence,
and prolonged post-inflammatory hyperpigmentation.
A definite causal relationship was established by Naranjo score (10-13), achieved through detailed clinical history and
two-episode temporal concordance, without requiring biopsy or provocative testing. Early recognition of FDE at the primary care level is essential to prevent unnecessary re-exposure, avoid potentially
more severe reactions, and ensure
safe substitution of the causative anti-inflammatory agent. Clinicians
should maintain
a high index of suspicion for drug- induced FDE when evaluating recurrent hyperpigmented lesions or oral erosions temporally associated with NSAID use.
The absence
of pharmacovigilance reporting
in this case highlights a persistent gap in primary care adverse drug reaction surveillance.
Healthcare providers in Peru are encouraged to report adverse
reactions including those to widely used COX-2 inhibitors through DIGEMID's national pharmacovigilance system, contributing to regional drug safety monitoring and evidence generation.
REFERENCIAS BIBLIOGRÁFICAS
1.
Porr C, Harris DM,
Vidrighin A, et al. Etoricoxib- induced
fixed erythema. J Clin Med. 2025;14(23):8504. doi:10.3390/jcm14238504
2.
Makris M, Papapostolou N,
Koumprentziotis IA, Pappa G, Katoulis
AC. Nimesulide-induced fixed drug
eruption followed by etoricoxib-induced fixed
drug eruption: an unusual case report and review of the
literature. J Clin Med. 2024;13(6):1583. doi:10.3390/jcm13061583
3.
Rawlings N, Joshi S, Sandison A, Carey B. Fixed drug eruption
secondary to etoricoxib. Br J Oral Maxillofac Surg. 2024;62(6):571-574. doi:10.1016/j.bjoms.2024.05.001
4. Perks A, Bates TJ, Velangi S, Brown RM, Poveda- Gallego A. Probable etoricoxib-induced
fixed drug eruption involving the
oral mucosa: a case report. Oral Surg
Oral Med Oral Pathol Oral Radiol. 2021;131(4):e100-e107.
doi:10.1016/j.oooo.2020.12.019
5. Roy SS, Mukherjee S, Era N, Mukherjee M. Etoricoxib-induced toxic epidermal
necrolysis: a fatal case report.
Indian J Pharmacol. 2018;50(3):139-142. doi:10.4103/ijp.IJP_39_17
6.
Khan DA, Banerji A,
Blumenthal KG, et al. Drug allergy: a 2022 practice
parameter update. J Allergy Clin Immunol. 2022;150(6):1333-1393. doi:10.1016/j.jaci.2022.08.028
7. Dextre-Vilchez, Rí´os-Lozano. Eritema medicamentoso pigmentado fijo por
etoricoxib. Iatreia. 2025.
doi:10.17533/udea.iatreia.280
8.
Siriwattanasatorn M,
Kamudhamas A, Sivapornpan N, Noonpugdee M, Kamalashiran C, Phetkate P. Fixed drug
eruption induced by etoricoxib in Thailand:
a case report. Toxicol Rep. 2025;14:102010.
doi:10.1016/j.toxrep.2025.102010
9. Mota D, Coutinho RM, Mesquita M, Cernadas J, Carneiro-Lea˜o L. Generalized fixed drug
eruption secondary to etoricoxib. Dermatitis. 2025;36(4):e405-e406.
doi:10.1089/derm.2024.0324.
10. Clinical and Research
Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and
Kidney Diseases; 2012-.
Adverse Drug Reaction
Probability Scale (Naranjo) in
Drug Induced Liver Injury. [Updated
2019 May 4]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK548069.
11. Naranjo CA, Busto U, Sellers EM, et al. A
method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30(2):239-245. doi:10.1038/clpt.1981.154.
12.
Sousa AS, Gonçalo M. Fixed
drug eruption by etoricoxib confirmed
by patch test. An Bras Dermatol. 2016;91(6):777-779. doi:10.1590/abd1806-4841.20164777
13.
Martí´nez Anto´n MD, Gala´n
Gimeno C, Sa´nchez de Vicente J,
Ja´uregui Presa I, Gamboa Setie´n PM. Etoricoxib-induced
fixed drug eruption: Report of seven
cases. Contact Dermatitis. 2020 Aug 18; doi:
10.1111/cod.13659