 |
INTRODUCTION
Comparative assessment of nutrient profiles and some essential trace metals in smoked Clarias gariepinus (Burchell, 1822) and Oreochromis niloticus (Linnaeus, 1758) from Owena River, Ondo State, Nigeria
Evaluación comparativa del perfil nutricional y de algunos oligoelementos esenciales en Clarias gariepinus (Burchell, 1822) y Oreochromis niloticus (Linnaeus, 1758) ahumados, procedentes del río Owena, estado de Ondo, Nigeria
E. T. Adebayo1*; P. O. Ayeku1; P. E. Kadiri1; C. Ogueri2; W. O. Adetunji-Popoola1
1 Department of Biosciences and Biotechnology, Faculty of Science, University of Medical Sciences, Ondo, Nigeria.
2 Department of Fisheries and Aquaculture Technology, School of Agriculture and Agricultural Technology, Federal University of Technology, Owerri, Nigeria.
* Corresponding Author: tadebayo@unimed.edu.ng (E. T. Adebayo).
ORCID of the authors:
E. T. Adebayo: https://orcid.org/0009-0004-2675-0325 P. O. Ayeku: https://orcid.org/0000-0001-5442-9660
ABSTRACT
This study evaluates the nutrition and some essential trace metal composition of smoked Oreochromis niloticus and Clarias gariepinus from the upper Owena River, Nigeria, in order to demonstrate the safety of these species for human consumption. The target hazard quotient (THQ) and estimated daily metal intake (EDMI) values of several macro- and micro-constituents were computed. The protein value of Clarias gariepinus was somewhat higher than that of Oreochromis niloticus, from 66% to 75% across the three body sections (fillet, skin, and gills). The result showed that potassium (249.65 ± 17.82 - 286.48 ± 13.14 mg/100 g), Sodium (39.13 ± 4.48 – 62.98 ± 2.41 mg/100 g), and Calcium (18.72 ± 1.19 - 30.58 ± 1.83 mg/100 g) are the most abundant mineral in both samples (C. gariepinus and O. niloticus). The trace metal concentration was K > Na > Ca > Zn > Fe > Cu > Al. The EDMI-measured levels of each metal in two fish species were higher than the range for daily allowance of metals per day per consumer weighing an average of 52 kg. Seven elements in O. niloticus and C. gariepinus have THQ values below one. Immune and digestive health is improved by vitamin A (12.95 – 13.22 g/100 g) and vitamin C (0.94 – 1.13 g/100 g). With low carbohydrate content and moderate cholesterol levels, the two species demonstrated the potential health benefits depending on dietary goals.
Keywords: keyword Nutrition; Consumption; Target Hazard Quotient (THQ); Immune.
RESUMEN
Este estudio evalúa el valor nutricional y la composición de algunos oligoelementos esenciales en Oreochromis niloticus y Clarias gariepinus ahumados, provenientes del río Owena (Nigeria), con el fin de demostrar la seguridad de estas especies para el consumo humano. Se calcularon el cociente de riesgo objetivo (THQ) y la ingesta diaria estimada de metales (EDMI) para varios macronutrientes y micronutrientes. El contenido de proteínas en Clarias gariepinus fue ligeramente mayor que en Oreochromis niloticus, entre 66% y 75% en las tres partes analizadas (filete, piel y branquias). Los resultados mostraron que el potasio (249,65 ± 17,82 - 286,48 ± 13,14 mg/100 g), el sodio (39,13 ± 4,48 – 62,98 ± 2,41 mg/100 g) y el calcio (18,72 ± 1,19 - 30,58 ± 1,83 mg/100 g) son los minerales más abundantes en ambas especies. La concentración de oligoelementos fue: K > Na > Ca > Zn > Fe > Cu > Al. Los niveles de metales en ambas especies fueron superiores a la ingesta diaria recomendada para una persona de 52 kg. Siete elementos en O. niloticus y C. gariepinus presentaron valores de THQ inferiores a 1. La vitamina A (12,95 – 13,22 g/100 g) y la vitamina C (0,94 – 1,13 g/100 g) contribuyen a la salud del sistema inmunitario y digestivo. Con bajo contenido de carbohidratos y niveles moderados de colesterol, ambas especies presentan beneficios para la salud, dependiendo de las necesidades nutricionales.
Palabras clave: Nutrición; Consumo; Cociente de riesgo objetivo (THQ); Sistema inmunitario.
Received: 24-03-2025.
Accepted: 10-09-2025.
A staple food around the world, fish is prized for its ability to supply vital elements such as healthy fats, vitamins, minerals, and high-quality proteins (Adebayo et al., 2016; Singh et al., 2025). Good protein, vitamins A, D, and B, vital minerals Fe, Zn, P, Ca, and I, and essential fatty acids, particularly long-chain polyunsaturated fatty acids (LCPUFA), can all be found in fish (Yetunde, 2016). The nutritional profiles of fish vary by species, habitat, and techniques of processing and preparation. Numerous health advantages, such as a decreased risk of cardiovascular disease, better maternal health and pregnancy outcomes, and enhanced physical and cognitive development in early infancy, have been associated with micronutrients found in fish (Tørris et al., 2018, Abraha et al., 2018; Noreen et al., 2025). Additionally, they enhanced immune system performance and reduced the risk of stunting, rickets, anemia, and childhood blindness brought on by micronutrient deficiencies (Ayeloja et al., 2013). Lysine, an essential amino acid that is low in cereals, is found in good concentration in fish (Gunarathne et al., 2025). As a result, fish protein can enhance a varied diet's overall protein quality and amino acid composition
Clarias and Tilapia are among the most popular freshwater fish species in Nigeria, due to their of their availability, economic significance, and rich nutritional profiles. This is especially true in areas like Ondo State, where local fisheries are essential to both food security and revenue development (Fagbenro et al., 2010). It is impossible to overstate the significance of catfish to the Nigerian populace. In addition to providing meals for the general public, it enables better protein nutrition in terms of the body's high protein retention. According to Olaoye et al. (2016), catfish is a fantastic source of omega-3 fatty acids and one of the safest animal protein sources. A common estuary cichlid on Africa's west coast, Tilapia guineensis is a significant fish species because it significantly contributes to the nutritional, growth, and development requirements of many African nations. It has good aquaculture, economic, and nutritional advantages, particularly when it comes to West Africans' protein intake (Ukenye et al., 2019).
Trace metals, which are present in minimal quantities in living tissues, are categorized into essential and non-essential metals (Alloway, 2013). Essential trace elements, such as iron, calcium, potassium, magnesium, zinc, sodium, phosphorus, copper, iodine, and selenium, are vital for numerous physiological functions, including enzymatic reactions and maintaining electrolyte balance (Rolić et al., 2025). The amount of key metals, especially sodium, potassium, calcium, and magnesium, in fish is directly related to its nutritional value. According to Adeyemi et al. (2015), these metals are essential for sustaining regular body processes like bone health, muscular contraction, and nerve transmission. However, fish health and, by extension, human consumers are at danger when non-essential metals are present in aquatic ecosystems due to industrial effluents, agricultural runoff, and other human activities. Fish bioaccumulate metals over time as a result of consuming polluted food or absorbing them through their gills. The rate of metal absorption is further influenced by variables such fish eating habits, sediment composition, and water pH (Ajani et al., 2022).
Due to its high protein content, omega-3 fatty acids, and minerals, which support brain function, cardiovascular health, and general well-being, eating fish has several health benefits (Olaoye et al., 2016). However, to ensure that they meet nutritional needs without posing health risks, a consistent investigation of the levels of essential metals, such as sodium, potassium, and calcium, in these fish is required to add to their baseline information and likely identify a shift in their value due to the community's adopted processing method. According to research, environmental issues like pollution and water quality have a substantial impact on the nutritional makeup of fish species (South et al., 2025). Adebayo et al. (2016) emphasized the impact of water chemistry on metal concentrations in fish tissues, whereas Adeyeye (2016) documented variation in the mineral content of fish species according to their habitats. This study therefore evaluates the nutritional composition and essential metal content of smoked-dried Clarias (O. gariepinus) and Tilapia (O. niloticus) species from the Owena River to provide insight into the safety and nutritional quality of fish consumed in the region, ensuring they remain a healthy and sustainable food source for the local population.
METHODOLOGY
Description of the study area
The Owena River is a significant river in southwestern Nigeria located between latitude 7°N to 6°N and longitude 5°E to 4°E. The river takes it source from Ekiti State where it flows southward through Ondo State where it empties into Ose River that drains into the Atlantic Ocean (Figure 1). The river serves as boundary between Osun State and Ondo State and plays a central role in agriculture, fishing, and other local economic activities that prompted indigenes from this duo state to form a settlement along its bank called Owena community in Igbara-Oke, Ifedore local government area.
Experimental design and analyses
Thirty (30) pieces of each smoked fish (Clarias gariepinus and Oreochromis niloticus) samples were purchased from the local market vendor who had freshly dried the fish on a designated market day.
Figure 1. Map of study area. (Source: Authors’ Download: Google map, 2018).
All the collected samples were transported to the laboratory, department of Biosciences and Biotechnology, University of Medical Sciences for thorough investigations and filleting of the tissues. The fish skins, fillets, and gills were sorted for nutritional value and some selected essential trace metal (sodium, calcium, potassium, iron, copper, aluminum and zinc) analysis. Proximate analysis was investigated in the Department of Biosciences and Biotechnology Laboratory, University of Medical Sciences, while metal analysis was examined in Chemistry Laboratory, Afe Babalola University, Ado-Ekiti, Ekiti State.
Proximate analysis
The AOAC (2007) standard procedures were used to determine the total fat, crude fiber, and carbohydrates. Every proximate analysis of the flour sample was performed three times and given as a percentage. Every chemical was of Analar grade.
Determination of crude protein
Using mercury (Hg) as a metal catalyst, the fish sample to be examined was digested using concentrated sulfuric acid, a little amount of copper sulfate, selenium, and enough sodium or potassium sulfate. Ammonium sulphate (NH4)2SO4 was produced because of the oxidation of organic materials and the nitrogen in the protein. After the digestion, a strong base (NaOH) was added to release ammonia. After distillation, the ammonia was trapped in a 0.5% boric acid indicator and titrated with 0.01 M HCl. The conditions of digestion transformed nearly all organic forms of nitrogen into ammonia. Crude protein is typically used to express the Kjeldahl analysis result. Based on the proportion of nitrogen in the protein sample, the weight of nitrogen in a sample can be converted to protein using the relevant factor. The common factors 6.25 were used to convert grams of nitrogen to grams of protein. Thus, the weight of protein was calculated by multiplying the nitrogen by 6.25 (Oladipo & Jadesimi, 2013).
Determination of mineral elements
The chemical analysis involved determination of the content of Na+, K+, Ca+2, Mg+2, P+2, Zn+2, and Cu+2 in mineralized fish meat samples. Following cremation at 480 degrees Celsius in a muffle furnace, the elements' contents in the fish materials (3 grams of fish meat) were ascertained. On crucibles, the resulting ash was dissolved with 6 mol l−1 of spectrally pure hydrochloric acid (POCH, Poland). Flame atomic emission spectroscopy (FAES) was used to investigate Na and K at wavelengths of λ = 589.0 nm and λ = 766.5 nm, respectively, using a flame photometer (Pye Unicam SP 2900, Cambridge, UK). Using a SOLAAR 939/959 spectrophotometer (Unicam, Cambridge, UK) and flame atomic absorption spectroscopy (FAAS), the elements Ca, Fe, Al, Zn, and Cu were identified. In accordance with Polish Norm PN-EN ISO 6869:2002 (2002), calcium was measured at λ = 422.7 nm, aluminium at λ = 285.2 nm, zinc at λ = 213.9 nm, and copper at λ = 324.8 nm.
An examination of approved reference materials for Multielement Trace Analysis Fish Tissue (IAEA-407, Atomic Energy Agency, Nutrients 2019, 11, 1448 4 of 12 Vienna) confirmed the analytical procedure's correctness. Relative standard deviations (RSD) and recovery levels for the elements under analysis were as follows: Cu (97.5%, 6.1%), Al (98.4%, 4.9%), Zn (97.3%, 6.8%), Ca (97.5%, 6.8%), K (99.2%, 5.3%), and Na (100.2%, 4.1%). Helios Alpha UV-VIS device (Spectronic Unicam, Leeds, UK) was used to determine the phosphorus concentration using the spectrometric method at 400 nm (AOAC, 2011)
Estimated daily metal intakes (EDMI)
Using the formula suggested by Kakar et al. (2020), the daily dose of metal intake was determined in order to assess the health hazards related to metal pollution in various fish species:
![]() (1)
(1)
where C is the average metal concentration in fish samples (micrograms per gram of fish flesh on a dried weight basis), BW is the average body weight of 52 kg for an adult human of the local population, as proposed by Kakar et al. (2020, and FIR is the fish ingestion rate (measured as 0.0306 kg/day in Nigeria according to Naylor et al., (2021).
Calculation of fish consumption data
Data on fish consumption is calculated: According to surveys conducted by the Food and Agriculture Organization (FAO) (2020) and the National Bureau of Statistics (Nigeria), the average annual consumption of fish among Nigerians is 11.2 kilograms per capital. The health risk assessment, including the non-carcinogenic and carcinogenic hazards of all these metals in fish species consumed by consumers at adult age, was evaluated using the target hazard quotient (THQ) values of the trace elements for intake by both fish species.
![]() (2)
(2)
where C is the trace metal concentration; ED is the length of exposure (for adults: 70 years); FIR is the consumption of each type of fish (0.0306 kg/person/day); For adults, EF stands for exposure frequency (365 days a year); BW for body weight (average of 52 kg); AT is average lifespan (10,950 days); and RfD for oral reference dose (FAO/WHO, 2019). The total target hazard quotient (TTHQ) was determined by adding the THQ of each individual chemical element using all of the trace element studies from each species of fish sample.
TTHQ = THQAl + THQNa + THQZn + THQFe + THQCu + THQK + THQCa (3)
If THQ or TTHQ was ≥ 1, consumers are at considerable health risk for these metals ingestion, but when THQ or TTHQ < 1, then is no non-carcinogenic health risk is associated with these fish consumptions.
Statistical analysis
SPSS Software version 20.0 and Microsoft Excel version 365 were used for all statistical analyses, including ANOVA and Tukey's multiple comparison tests at a significance level of p < 0.05.
RESULTS AND DISCUSSION
The basic nutritional composition of the dried Clarias gariepinus and Oreochromis niloticus used in this investigation were presented in Table 1 below. Crude protein content (CP) study showed that the three body components (fillet, skin, and gills) varied somewhat, ranging from 66% to 75%. Oreochromis niloticus has a lower CP value than Clarias gariepinus. There were minor variations in total fat content between the two fish species and within body parts, with values ranging from 2.84% in the O. niloticus fillet to 4.17% in the C. gariepinus skin. The total cholesterol levels of the two fish analyzed in this investigation showed a similar pattern. In contrast, O. niloticus had substantially more vitamin A and dietary fiber than C. gariepinus. Vitamin B12, Vitamin C and Carbohydrate quantities follow similar trends between the sampled fish species and across the body parts with no significant variations observed. The high protein content that was observed supported the claim made by Ahmed et al. (2011) that fish is a high-quality protein source that is necessary to produce enzymes, the maintenance of immunological function, and the construction and repair of bodily tissues. Fish proteins are complete because they include every necessary amino acid needed by the human body (Ravichandran et al., 2011). Tilapia and Clarias are both known to have a high protein content, which makes them an excellent source of dietary protein in areas like Ondo State where fish is a staple food. With a high percentage of peptides and essential amino acids, which are rare in meat proteins from other sources, fish constitute a significant supply of high-quality, easily digestible proteins. The building blocks of muscles, hair, and other body parts, proteins are a vital supply of enzymes and antibodies (Rasul et al., 2021).
High amounts of n-3 polyunsaturated fatty acids (PUFA), such as docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA), which have numerous health advantages, are found in fish lipids (Glover-Amengor et al., 2014).
Table 1
Proximates analyses of Clarias gariepinus and O. niloticus from Owena River, Igbara-Oke, Ondo State, Nigeria
|
Species/ Parameters |
C. gariepinus |
O. niloticus |
||||
|
Fillet |
Skin |
Gill |
Fillet |
Skin |
Gill |
|
|
Total Fat- TF (%) |
3.40±0.21 |
4.17±0.08 |
3.20±0.10 |
2.84±0.14 |
2.88±0.47 |
3.52±0.29 |
|
Crude Protein - CP (%) |
77.16±2.00 |
73.21±1.68 |
75.33±1.90 |
71.13±1.32 |
71.25±3.02 |
69.46±0.73 |
|
Total Cholesterol-TC (%) |
1.11±0.03 |
1.08±0.06 |
1.15±0.02 |
0.97±0.02 |
1.01±0.04 |
1.00±0.09 |
|
Dietary Fiber - DF (g) |
2.26±0.09 |
2.40±0.07 |
2.43±0.10 |
2.78±0.08 |
2.71±0.08 |
2.75±0.13 |
|
Vitamin A - VA (IU) |
11.98±0.54 |
12.20±0.12 |
12.00±0.24 |
12.95±0.80 |
13.07±0.40 |
13.22±0.21 |
|
VB12 (g) |
0.34±0.08 |
0.32±0.02 |
0.34±0.05 |
0.30±0.08 |
0.34±0.04 |
0.30±0.02 |
|
Vitamin C - VC (g) |
0.90±0.07 |
0.92±0.04 |
0.88±0.07 |
0.94±0.04 |
1.13±0.05 |
0.98±0.02 |
|
Carbohydrate - CHO (g) |
0.01±0.00 |
0.01±0.01 |
0.02±0.01 |
0.03±0.00 |
0.02±0.00 |
0.03±0.01 |
EPA and DHA have been shown to improve cardiovascular health, prevent arthritis, inflam-mation, aging, and insulin resistance (Pal et al., 2019). Furthermore, the essential fatty acids present in fish oils have been shown to reduce depression, slow the progression of many cancers, and promote children's and adolescents' cognitive development (Kwasek et al., 2020). In contrast to the findings of Sultna et al. (2011), who found lipid contents in seven SIS dried fishes (12.20 percent to 22.70 percent), the total crude fat level of C. gariepinus and O. niloticus in this investigation was substantially low. The two fish species in this study can be categorized as low-fat fish based on the results obtained (having less than 8% fat contents) following the Ackman (1989) grouping methods.
The mineral compositions of O. niloticus and C. gariepinus in milligrams per 100 grams were presented in Table 2. The findings indicated that the most prevalent minerals in both samples (C. gariepinus and O. niloticus) were potassium (249.65±17.82 - 286.48±13.14 mg/100 g), sodium (39.13±4.48 - 62.98±2.41 mg/100 g), and calcium (18.72±1.19 - 30.58±1.83 mg/100 g). There was a range of 1.65±0.15 to 3.02±0.20 mg/100 g for iron and 0.18±0.04 and 0.49±0.26 mg/100 g for aluminum in the samples. Of the two fish species that were sampled, the trace element zinc was substantially greater than Cupper. According to Adeyeye (2014) and Njinkoue et al. (2016), potassium is the most prevalent mineral in fish species, which is consistent with this finding.
Variability in the accumulation of mineral elements in tissues, nutrient availability in the aquatic environment or feed, and the capacity to absorb and convert the compounds into vital nutritional components are the main causes of the variations in nutrient concentrations seen in the fish species under analysis (Fawole et al., 2007). Fish habitat (whether bottom-dwelling or surface-feeding), feeding habits (whether herbivorous, carnivorous, or omnivorous), the kind of pollutants in their aquatic environment, fish size, metabolic rate, and biotransformation capacity are some of the variables that affect the buildup of trace elements and metalloids in fish muscles (Santhana et al., 2022). For example, freshwater fish's gills, which are rich in iron, allow them to absorb large amounts of iron from the water.
Among the metals examined, potassium (K) showed the highest value, and the trace metal concentration was found to be K > Na > Ca > Zn > Fe > Cu > Al. The observed differences in trace metal accumulation in fish samples could be explained by a number of factors, such as the fact that fish samples taken from locations near pollution sources may have different metal concentrations or different metabolic rates or efficiencies in accumulating and removing metals from their bodies, which could result in different metal concentrations between the two fish species. Additionally, metals in fish tissues may interact with other organic materials, such protein, which may make them more difficult to identify using analytical techniques. According to a number of studies (Nazeer et al., 2023; Mehdi-Hosseini et al., 2022), fish exposed to high levels of waterborne metals have the ability to absorb or consume contaminated food and water, which causes the metals to bioaccumulate through their skin, gills, and digestive tract. Therefore, sediment composition, water quality changes, or the quantity of contaminated water or food consumed are the main factors linked to the metal concentration levels in each fish sample. Each of these elements has a part in the fluctuations in metal detection found in fish samples.
C. gariepinus and O. niloticus have total cholesterol levels that indicate a greater preference for adult meals in order to prevent cardiovascular disease. The findings have also demonstrated the two species' vitality in the diets of humans and animals for the best possible growth and development. Additionally, this study demonstrated that tilapia and clarias are significant suppliers of the vitamins A, B12, and C that are necessary for sustaining human health. Vitamin A (retinol) promotes healthy skin, eyesight, and the immune system. Given their high vitamin A content, fish like Clarias are an excellent nutritional choice, especially for people at risk of vitamins deficiency. Vitamin B12 (Cobalamin), is essential for the production of red blood cells and for the health of the nervous system. As previously noted by Chukwu and Shaba (2009), both Tilapia and Clarias are great providers of B12. However, because of their diet, Clarias have higher levels of B12. Fish typically have low levels of vitamin C (ascorbic acid), which helps with collagen synthesis and iron absorption. However, because of the nutritional feed, farmed fish can have higher levels of vitamin C (Sadiku & Oladimeji, 1991).
Table 2
Trace metals concentrations of C. gariepinus and O. niloticus from Owena River, Ondo State Nigeria
|
|
C. gariepinus |
O. niloticus |
|
||||
|
Fillet |
Skin |
Gill |
Fillet |
Skin |
Gill |
||
|
Na |
46.98±0.44 |
52.90±2.35 |
62.98±2.41 |
47.10±3.61 |
39.13±4.48 |
50.77±7.36 |
|
|
Ca |
23.92±1.84 |
30.58±1.83 |
26.92±2.28 |
20.98±0.55 |
18.72±1.19 |
24.80±1.31 |
|
|
K |
249.65±17.82 |
286.48±13.14 |
470.28±11.44 |
276.62±15.21 |
251.22±14.9 |
271.27±5.8 |
|
|
Fe |
1.93±0.13 |
2.00±0.18 |
1.65±0.15 |
1.73±0.37 |
3.01±0.15 |
3.02±0.20 |
|
|
Zn |
2.11±0.08 |
2.60±0.57 |
1.94±0.06 |
2.19±0.36 |
2.18±0.08 |
2.07±0.07 |
|
|
Cu |
0.72±0.17 |
0.91±0.22 |
0.91±23 |
0.55±0.32 |
1.00±0.46 |
1.02±0.46 |
|
|
Al |
0.44±0.26 |
0.18±0.04 |
0.19±0.04 |
0.45±0.36 |
0.49±0.26 |
0.48±0.23 |
|
The two fish species (C. gariepinus and O. niloticus) had their estimated daily metal intake (EDMI) values in mg/person/day shown in Figure 2. Overall, the results indicated that the EDMI-measured values of each metal analyzed in the two fish species of the Owena River were found to be above the minimum recommended daily allowance range of metals in mg/day/person for consumers with an average weight of 52 kg (FAO 1983; USEPA 1989). As a result, these species can meet the daily metal intake requirements of humans. The freshwater species examined in the study had high levels of K, Ca, Na, and Zn, while Cu showed low levels. Since 1990s, studies on the relationship between consumption, balance, and biomarkers of copper status in the French population have shown that extremely low daily intakes (less than 0.8 mg/day) might result in copper losses and have long-term negative health effects. These analysis' findings demonstrate that consumers' long-term health benefits are guaranteed by their consumption of copper. The development of the dietary Cu requirement is hampered by the authors' warning that high Cu concentrations are hazardous (Bost et al., 2016).
The target hazard quotient (THQ) values were calculated in this study to assess the possible harm that trace metals in fish could cause to people through the intake of food contaminated with them (Figure 3). Eating such meals has no immediate negative health impacts if THQ is less than 1. However, THQ values of the seven elements examined in O. niloticus and C. gariepinus were less than one (THQ < 1) in our current investigation. Therefore, eating these two fish species has no negative consequences on human health as a result, metal deposition in bodily tissues is dose-dependent and correlated with exposure time, mirroring environmental metal concentrations. The potential health risk posed by these chemical contaminants is dependent not only on the amount of metals consumed but also on the frequency and duration of exposure, the oral reference dose (RfD), and the average body weight of the consumers, according to Javed and Usmani's (2016) study on the accumulation of heavy metals and the assessment of human health risk through the consumption of freshwater fish. Furthermore, it was mentioned that THQ represents the degree of concern in addition to being a gauge of health risk.
Minerals are necessary for the control of bodily functions such muscle contraction, nerve transmission, and bone formation. Essential elements such as calcium, phosphorus, magnesium, iron, and zinc—all necessary for sustaining human health—can be found in abundance in fish (Javed and Usmani's (2016). The essential metal's mild concentration in the reported samples (Table 2) complies with the FAO/WHO (2019) regulatory limits for safe human consumption.
Even so, the smallest amount of heavy metal is frequently used to quantify the muscles in fish bodies. Because muscles are used in the human diet, they are typically given attention. According to earlier research, calcium is a plentiful mineral that the body requires for normal growth and repair of the muscles and central nervous system (CNS). In addition to being essential for blood clotting, calcium is stored in bones and teeth. Low calcium levels can cause osteomalacia and calcium rickets (Khawar et al., 2024). The majority of enzymes, which are involved in the anabolic and catabolic pathways, require zinc to function correctly. Copper is a fantastic cofactor that is essential as a metabolite and aids in the production of hemoglobin (Khawar et al., 2024). All of the trace metals identified in this study are of great significance since they are required for the normal functioning of the human body, which is vital for survival. Overall, this study highlights the nutritional worth of these commercially significant freshwater fish species from the Owena River and their contribution to a balanced diet.
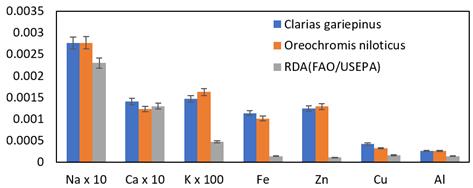
Figure 2. EDMI of C. gariepinus and O. niloticus from Owena River, Ondo State Nigeria.
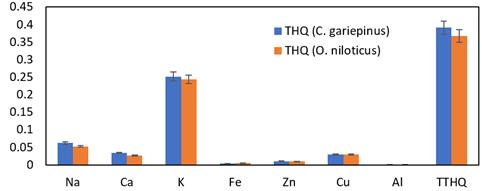
Figure 3. THQ and TTHQ of C. gariepinusand O. niloticus from Owena River, Ondo State Nigeria.
CONCLUSIONS
In conclusion, C. gariepinus and O. niloticus can meet the daily metal intake requirements of humans. The freshwater species analyzed in the study had a high K, Ca, Na, and Zn level while Cu exhibited low amount. The study also highlights the high protein content and slight variations in crude protein values between C. gariepinus and O. niloticus from the Owena River. The two fish species do not exhibit any negative health effects when consumed. There are a number of potential reasons for the variations in trace metal accumulation in the two freshwater fish samples examined in this study, including fish samples taken from locations near pollution sources and different metabolic rates. By understanding and controlling these variables, it is feasible to guarantee the sustainability and safety of fish as a wholesome food source while reducing any potential health hazards for consumers. It is therefore recommended that a regular monitoring of heavy metal concentrations in O. niloticus and C. gariepinus from Owena River to ensure that fluctuating environmental conditions do not elevate toxic metal levels above safe thresholds.
REFERENCES
Abraha, B., Admassu, H., Mahmud, A., Tsighe, N., Shui, X. W., & Fang, Y. (2018). Effect of processing methods on nutritional and physico-chemical composition of fish: a review. MOJ Food Process Technol, 6(4), 376–382. http://doi.org/10.15406/mojfpt.2018.06.00191
Ackman, R. G. (1989). Nutritional composition of fats in seafoods. Prog Food Nutr Sci, 13(3-4), 161-289.
Adebayo, I. A., Fapohunda, O. O. & Ajibade, A. O. (2016). Evaluation of nutritional quality of Clarias gariepinus from selected fish farms in Nigeria. American Journal of Food Science and Nutrition Research, 3(4), 56-62.
Adeyemi, B. R., Adeseye, S. M., Olajuyigbe, O., & Akande, G. (2015). Proximate and Mineral Composition in the Flesh of Five Commercial Fish Species in Nigeria. Internet Journal of Food Safety, 13, 208 - 213.
Adeyeye, E. I. (2016). Determination of Heavy Metals in Fish from Some Local Ponds in Ekiti State. Journal of Environmental Chemistry and Ecotoxicology, 5(3), 99-107.
Ahmed, I., Jan, K., Fatma, S., & Dawood, M. A. O. (2022). Muscle proximate composition of various food fish species and their nutritional significance: A review. Jour of Animal Physio and Animal Nutri, 106, 690–719. https://doi.org/10.1111/jpn.13711
Ajani, M. B., Maleka, P. P., Penabei, S., & Usman, I. T. (2022). Health risk assessment of heavy metals concentration from soil; a case study of the Mayo-Dallah in Southern area of Chad. Journal of Radiation Research and Applied Sciences, 15(1), 130-138. https://doi.org/10.1016/j.jrras.2022.01.013
Alloway, B. (2013). Heavy Metals in Soils: Trace Metals and Metalloids in Soils and their Bioavailability. Book Springer Nature. https://doi.org/10.1007/978-94-007-4470-7
AOAC. Official Methods of Analysis of AOAC International, 18th ed.; Revision 4; Horwitz, W., Latimer, G.W., Jr.,Eds.; AOAC International: Gaithersburg, MA, USA, 2011.
AOAC. Official Methods of Analysis. Assoc Off Anal Chem. 18th Edit; 2007.
Ayeloja, A. A., George, F. O. A., Dauda, T. O., Jimoh, W. A., & Popoola, M. A. (2013). Nutritional comparison of captured Clarias gariepinus and Oreochromis niloticus. Int Res J Nat Sci, 1(1), 9–13. https://doi.org/10.37745/irjns.13
Bost, M., Houdart, S., Oberli, M., Kalonji, E., Huneau, J. F., & Margaritis, I. (2016). Dietary copper and human health: Current evidence and unresolved issues. J. Trace Elem. Med. Bio, 5, 107–115.
Chukwu, O., & Shaba, I. M. (2009). Effects of Drying Methods on Proximate Compositions of Catfish (Clarias gariepinus). World Journal of Agricultural Sciences, 5, 114-116.
Fagbenro, O. A., Jegede, T., & Fasasi O. S. (2010). Tilapia Aquaculture in Nigeria. Applied Tropical Agriculture, 15, 49-55.
FAO (Food and Agricultural Organization) (1983). Compilation of legal limits for hazardous substances in fish and fishery products. FAO Fishery Circular No. 464. Food and Agriculture Organization, pp. 5–100.
FAO. (2020). New Food Balances. http://www.fao.org/faostat/en/#data/FBS
Fawole, O. O., Ogundiran, M. A. Ayandiran, T. A., & Olagunju, O. F. (2007). Mineral composition in some selected fresh water fishes in Nigeria. J. Food Safety, 9, 52–55.
Food and Agriculture Organization of the United Nations. (2019). Dietary intake of vitamins and minerals: FAO/WHO expert consultation on human vitamin and mineral requirements. Food and Agriculture Organization of the United Nations. Retrieved from http://www.fao.org/3/y2809e/y2809e00.htm
Glover-Amengor, M., Atikpo, O., Abbey, L. D., Hagan, L., Ayin, J., & Toppe, J. (2012). Proximate Composition and Consumer Acceptability of Three Underutilized Fish Species and Tuna Frames. World Rural Obs, 4(2), 65–70.
Gunarathne, R., Guan, X., Feng, T., Zhao, Y., & Lu, J. (2025). L-lysine dietary supplementation for childhood and adolescent growth: Promises and precautions. J Adv Res., 70, 571-586. http://doi.org/10.1016/j.jare.2024.05.014
Javed, M., & Usmani, N. (2016) Accumulation of heavy metals and human health risk assessment via the consumption of freshwater fish Mastacembelus armatus inhabiting, thermal power plant effluent loaded canal. Springer Plus, 5(1), 776. https://doi.org/10.1186/s40064-016-2471-3
Kakar, A., Hayat, M. T., Abbasi, A. M., Pervez, A., Mahmood, Q., Farooq, U., Akbar, T. A., Ali, S., Rizwan, M., El-Serehy, H. A., & Abdel-Daim, M. M. (2020). Risk Assessment of Heavy Metals in Selected Marine Fish Species of Gadani Shipbreaking Area and Pakistan. Animals, 10(10), 1738. https://doi.org/10.3390/ani10101738
Khawar, M., Masood. Z., Hasan, H. U, Khan, W., De los Ríos‑Escalante, P. R., Aldamigh, M. A., Al‑Sowayan, N. S., Razzaq, W., Khan, T., & Said, M. B. (2024). Trace metals and nutrient analysis of marine fish species from the Gwadar coast. Scientific Reports, 14, 6548. https://doi.org/10.1038/s41598-024-57335-0
Kwasek, K., Thorne-Lyman, A. L., & Phillips, M. (2020). Can human nutrition be improved through better fish feeding practices? a review paper. Crit Rev Food Sci Nutr, 60(22), 3822-3835. http://doi.org/10.1080/10408398.2019.1708698
Law Journal. UE L 304, 2011. Regulation (EU) No 1169/2011 of the European Parliament and of the Council of 25 October 2011 on the provision of food information to consumers, amending Regulations (EC) No 1924/2006 and (EC) No 1925/2006 of the European Parliament and of the Council and repealing Commission Directive 87/250/EEC, Council Directive 90/496/EEC, Commission Directive 1999/10/EC, Directive 2000/13/EC of the European Parliament and of the Council, Commission Directives 2002/67/EC and 2008/5/EC and Commission Regulation (EC) No. 608/2004, Annex XIV “Conversion Rates”.
Mehdi-Hosseini, S., et al. (2022). Risk assessment of trace elements bioaccumulated in golden gray mullet (Liza aurata) harvested from the southern Caspian Sea. J. Gt. Lakes Res., 48(4), 1079–1086. https://doi.org/10.1016/j.jglr.2022.04.010
Naylor, R. L., Kishore, A., Sumaila, U. R., Issifu, I., Hunter, B. P., Belton B., Bush, S. R., Cao, L., Gelcich, S., Gephart, J. A., Golden, C. D., Jonell, M., Koehn, M. Z., Little, D. C., Thilsted, S. H., Tigchelaar, H., & Crona, B. (2021). Blue food demand across geographic and temporal scales. Nature communications, 12, 5413. https://doi.org/10.1038/s41467-021-25516-4
Nazeer, N., et al. (2023) Impacts of some trace metals in Cyprinus carpio (Linnaeus, 1758) and Tor soro (Valenciennes, 1842) on human health. Biol. Trace Elem. Res. https://doi.org/10.1007/s12011-023-03852-4
Noreen, S., Hashmi, B., Aja, P. M., & Atoki, A. V. (2025) Health benefits of fish and fish by-products-a nutritional and functional perspective. Front Nutr, 9(12), 1564315. http://doi.org/10.3389/fnut.2025.1564315
Oladipo I. C., & Jadesimi P. D. (2012). Microbiological analysis and nutritional evaluation of West African soft cheese (wara) produced with different preservatives. American Journal of Food Nutrition, 3(1), 13-21. http://doi.org/10.5251/ajfn.2013.3.1.13.21
Olaoye, J., Ezeri, G., Akegbejo-Samsons, Y., Awotunde, J., & Ojebiyi, W. (2016). Dynamics of the adoption of improved aquaculture technologies among fish farmers in Lagos State, Nigeria. Croatian Journal of Fisheries, 74, 56-70. http://doi.org/10.1515/cjf-2016-0012
Pal, J., Shukla, B. N., Maurya, A. K., & Verma, H. O. (2018). A review on role of fish in human nutrition with special emphasis to essential fatty acid. Int J Fish Aquat Stud, 6(2), 427-430.
PN-EN ISO 6869. Animal Feeding Stuffs—Determination of the Contents of Calcium, Copper, Iron, Magnesium, Manganese, Potassium, Sodium and Zinc—Method Using Atomic Absorption Spectrometry; Polish Committee for Standardization: Warsaw, Poland, 2002.
Rasul, M. G., Yuan, C., & Shah, A. K. (2021). Chemical composition and nutritional value of dried fish in Bangladesh. Egypt J Aquat Biol Fish, 25(4), 379–399. http://doi.org/10.21608/EJABF.2021.189666
Ravichandran, S., Kumaravel, K., & Florence, E. P. (2011). Nutritive composition of some edible fin fishes. Inter J. of Zoological Res, 7(3), 241‐251. https://doi.org/10.3923/ijzr.2011.241.251
Rolić, T., Yazdani. M., Mandić, S., & Distante, S. (2025). Iron Metabolism, Calcium, Magnesium and Trace Elements: A Review. Biol Trace Elem Res. 203(4), 2216-2225. http://doi.org/10.1007/s12011-024-04289-z
Singh, R. D., Arambam, K., Singh, A. M., & Singh, S. K. (2025). Health and Nutritional Benefits of Fish as Human Food. In: Sundaray, J. K., Rather, M. A., Ahmad, I., Amin, A. (eds) Food Security, Nutrition and Sustainability Through Aquaculture Technologies. Springer, Cham. https://doi.org/10.1007/978-3-031-75830-0_3
South, J., Sabini, L., Pattison, Z., & Cuff, J. P. (2025). Aquatic biological invasions exacerbate nutritional and health inequities, Trends in Ecology & Evolution, 40(8), 718-721. https://doi.org/10.1016/j.tree.2025.06.00
Sadiku, S. O. E., & Oladimeji, A. A. (1991). Relationships of proximate composition of Lates niloticus Synodontis schall (brochand schnider) and Sarotherodon galiaeus (trewavas) from Zaria dam, Nigeria. Bioscience Research Community, 3, 29-40.
Sultana, S., Parween, S., & Hossain, M. A. (2011). Biochemical analysis of some dried sis fishes of the river Padma in Rajshahi. J Life Earth Sci, 6, 39–43. http://doi.org/10.3329/jles.v6i0.9719
Tørris, C., Småstuen, M. C., & Molin, M. (2018). Nutrients in fish and possible associations with cardiovascular disease risk factors in metabolic syndrome. Nutrients, 10(7), 952. http://doi.org/10.3390/nu10070952
U.S. Food and Drug Administration. (2016). Sodium in your diet. U.S. Department of Health and Human Services. https://www.fda.gov/food/food-labeling-nutrition/sodium-your-diet
Ukenye, E. A., Taiwo, I. A., & Anyanwu, P. E. (2019). Morphological and genetic variation in Tilapia guineensis in West African coastal waters: A mini review. Biotechnol Reports, 24, e00362. http://doi.org/10.1016/j.btre.2019.e00362
USEPA (United States Environmental Protection Agency) (1989). Office of Water Regulations and Standard: Guidance manual for assessing human health risks from chemically contaminated, fish and shellfish U.S. Environmental Protection Agency, Washington, DC;EPA-503/8-89-002.
World Health Organization. (2021). Guidelines for drinking-water quality: Fourth edition incorporating the first addendum. World Health Organization. https://www.who.int/publications/i/item/9789240062739
Yetunde, K. O. (2016). Biochemical composition of Micromesistus poutassou from agbalata market, Badagry Lagos West, Nigeria. Food Appl Biosci J, 4(1), 12-24. http://doi.org/10.14456/fabj.2016.2