 |
INTRODUCTION
Rootstocks as a strategy to prevent wilting in granadilla (Passiflora ligularis Juss.) cultivation
Portainjertos como estrategia para evitar la marchitez en el cultivo de granadilla (Passiflora ligularis Juss.)
Pablo Viteri1; Pablo Gaona2; Michelle Noboa3; Jorge Merino1*; Paúl Mejía1; Milton Hinojosa1;
William Viera1
1 Programa Nacional de Fruticultura, Instituto Nacional de Investigaciones Agropecuarias (INIAP), Eloy Alfaro y Av. Interoceánica, Tumbaco, Ecuador.
2 Consultor independiente, Quito, Ecuador.
3 Universidad Santiago de Compostela, Programa de Doctorado en Agricultura y Medioambiente para el Desarrollo, Santiago de Compostela, España.
* Autor corresponsal: jorge.merino@iniap.gob.ec (J. Merino).
ORCID de los autores
P. Viteri: http://orcid.org/0000-0003-3119-5798 P. Gaona: http://orcid.org/0000-0002-0606-5855
M. Noboa: https://orcid.org/0000-0002-2497-4702 J. Merino: http://orcid.org/0000-0002-5632-2194
P. Mejía: http://orcid.org/0000-0002-6772-8608 M. Hinojosa: https://orcid.org/0009-0004-6530-6649
W. Viera: http://orcid.org/0000-0003-4472-4126
ABSTRACT
In South America, diseases caused by soil-borne pathogens such as Fusarium are a major limiting factor for subtropical crops such as passion fruit (Passiflora ligularis Juss). This study evaluated the response to the use of rootstocks from Passifloraceae species related to passion fruit. A survival analysis was conducted to assess the resistance of this fruit crop to wilt under field conditions in an inter-Andean region of northern Ecuador. P. ligularis plants grafted onto rootstocks of four genotypes were evaluated, including P. edulis f. edulis (gulupa), P. edulis f. flavicarpa (yellow passion fruit), P. quadrangularis (badea), P. alata (sweet passion fruit), and P. ligularis as control (ungrafted). The highest yields were observed in plants grafted onto P. edulis f. edulis and P. edulis f. flavicarpa with 23.27 kg plant-1 and 20.52 kg plant-1, respectively, over a 6-month harvest period. These rootstocks also had the highest plant survival rates (95.60% and 92.00%, respectively). Consequently, gulupa and yellow passion fruit genotypes could be used as rootstocks to prevent the effects of wilting in granadilla crops.
Keywords: Fusarium; grafting; yield; survival; soluble solids.
RESUMEN
En Sudamérica, las enfermedades causadas por patógenos del suelo como Fusarium son un limitante importante para los cultivos subtropicales como la granadilla (Passiflora ligularis Juss). En esta investigación se evaluó la respuesta del uso de portainjertos de especies de Passifloráceas emparentadas con la granadilla. Se realizó un análisis de supervivencia para evaluar la resistencia de este frutal a la marchitez en condiciones de campo en una región interandina del norte de Ecuador. Se evaluaron plantas de P. ligularis injertadas sobre portainjertos de 4 genotipos que comprenden las especies P. edulis f. edulis (gulupa), P. edulis f. flavicarpa maracuyá amarillo), P. quadrangularis (badea), P. alata (maracuyá dulce), y como testigo absoluto P. ligularis (sin injertación). Los rendimientos más altos se observaron en plantas injertadas en P. edulis f. edulis y P. edulis f. flavicarpa con 23,27 kg planta-1 y 20,52 kg planta-1, respectivamente en un periodo de cosecha de 6 meses. Igualmente, estos portainjertos presentaron los mayores porcentajes de sobrevivencia de plantas (95,60% y 92,00%, respectivamente). Consecuentemente, genotipos de gulupa y maracuyá amarilla podrían ser utilizados como portainjertos para evitar el efecto de la marchitez en el cultivo de la granadilla.
Palabras clave: Fusarium; injertación; rendimiento; sobrevivencia; sólidos solubles.
Recibido: 21-04-2025.
Aceptado: 09-09-2025.
The genus Passiflora of the family Passifloraceae comprises approximately 450 species, mainly distributed in tropical regions, and it is notable for its economic, medicinal, and ornamental value (Ayres et al., 2015). Several domesticated and wild Passifloraceae species grow in South America due to their nutritional and nutraceutical properties (Viera et al., 2022a, 2022b, 2025). However, only a few species are cultivated commercially, while the majority are wild and their fruits are consumed locally (Joy, 2010). A total of 165 species are reported in the Andean region above 1800 masl, such as those in the subgenus Tacsonia (taxo or curuba), and species of the subgenus Passiflora (granadilla, badea, gulupa), which are distributed from sea level up to 3500 masl (Escobar, 1991). Passiflora is the most diverse genus and the only one with endemic species in the country, comprising 94 native and 34 endemic species (Jorgensen & Tye, 2017).
The granadilla (P. ligularis Juss.) is native to the Andes Mountain range (García, 2008) and is among the products in high demand in both national and international markets. It is primarily marketed as a fresh fruit and highly appreciated for its organoleptic characteristics and nutritional composition, which has generated significant economic interest among growers in regions dedicated to its cultivation (Bermúdez & Rodríguez, 2009). This species ranks as the second most economically important within the Passiflora genus, after passion fruit (P. edulis f. flavicarpa), due to its pleasant flavor and nutritional value, which favor its commercialization in both domestic and international markets (Yockteng et al., 2011).
Currently, granadilla cultivation has gained importance in the northern region of Ecuador (Gaona-Gonzaga et al., 2020). This crop develops optimally at altitudes between 1800 and 2300 masl, with temperatures ranging from 16 to 24°C. It requires deep, well-drained loamy soils, tolerates moderate winds, and annual rainfall between 2000 and 2500 mm with good distribution (Arias et al., 2016; Espinosa et al., 2018). Granadilla requires at least 4 mm of water per plant per day during the rainy season and up to 9 mm during the dry season; thus, proportional management of water and humidity is essential for crop development (Castro, 2020). However, traditional irrigation methods such as “flood irrigation” can serve as a vector for soil-borne pathogens.
One major limitation for this fruit crop is soil diseases (Sierra-Escobar, 2017). Studies on this topic indicate that pathogens associated with wilting symptoms are related to Fusarium solani and nematodes such as Meloidogyne javanica (Hoyos-Carvajal & Castillo Corredor, 2015; Sierra-Escobar, 2017). These diseases exhibit different symptoms: F. solani mainly affects the collar region, while M. javanica causes root galling (Ortiz & Hoyos, 2009; Hoyos-Carvajal & Castillo Corredor, 2015).
A plant infected by F. solani under field conditions typically exhibits progressive symptoms such as yellowing, flaccidity, or foliar wilting, which are characteristic of the disease (Hoyos-Carvajal & Castillo Corredor, 2015). When the infection occurs in the central root or stem region, it may advance internally without any visible damage to the outer layers or wood bark. External symptoms appear later as the fungus ascends through the xylem, rapidly colonizing vascular bundles until the plant dies (Tamayo, 1999).
The F. solani species complex displays remarkable genetic variability and adaptation to different hosts, which complicates its control and management in agriculture. The subdivision into special forms and the presence of supernumerary chromosomes encoding virulence factors highlight
the need for specific and advanced approaches to control diseases (Martínez et al., 2024).
caused by these pathogens.
As a soil-borne vascular disease, Fusarium wilt is difficult to control through conventional methods; therefore, preventative measures are the primary strategy (Roncatto et al., 2004; Preisigke et al., 2015; Lima et al., 2017). However, variability in Fusarium wilt resistance has been observed in some wild Passiflora species (Bueno et al., 2014; Preisigke et al., 2015), suggesting that the use of rootstocks could be an effective measure.
The use of rootstocks has been reported as an effective practice to avoid the effect of Fusarium wilt in several horticultural and fruit species (Khai et al., 2025; Molagholizadeh et al., 2025; Namisi et al., 2025; Wang et al., 2025)
According to Meletti et al. (2005), in studies evaluating resistance in Passiflora species, several have been identified as resistant to F. oxysporum, including P. giberti, P. alata, and P. caerulea. In yellow passion fruit, the use of P. maliformis has been reported as a rootstock resistant to F. solani (Ocampo et al., 2022); while this species showed tolerance against F. oxysporum in purple passion fruit (López et al., 2023). In the case of granadilla, grafting has been carried out with gulupa (P. edulis), as this wild species exhibits greater tolerance to soil-borne phytopathogenic fungi responsible for vascular wilt or fusariosis (F. oxysporum) and “Secadera” or brown stem rot (F. solani) (Guerrero & Hoyos-Carvajal, 2011; ICA, 2011).
Due to the above-mentioned, the objective of this study was to evaluate four Passifloraceae genotypes as rootstocks to determine granadilla plant survival.
METHODOLOGY
Location of the experiment
The study was conducted under field conditions at the site known as "6 de Julio" in the Parish of Cuellaje, Cotacachi County, Imbabura Province; located at coordinates 0°24'8.9" N, 78°24'5.7" W and an altitude of 1890 masl. The site has an average annual precipitation of 2250 mm with a bimodal interannual distribution pattern, an average annual temperature of 17 °C, and a relative humidity of 85%. The climate in this area corresponds to a semi-humid meso-thermal equatorial type.
Plant material
To initiate the trial, seedbeds were prepared using a commercial granadilla variety and different Passifloraceae rootstocks. All genotypes were obtained from production farms and local markets.
After 45 days, the seedlings were transplanted into bags, and grafting was performed once the rootstocks reached 0.5 cm in diameter (Rivera et al., 2002), and both the rootstock and scion had developed four to five true leaves (Santos et al., 2016a; Lima et al., 2017). The grafting method used was terminal cleft grafting. Field transplantation was carried out when the grafted granadilla plants reached approximately 0.25 m in height and had at least five functional leaves (Rivera et al., 2002). Planting distances for the trial setup were 5.0 m between rows and 5.0 m between plants, resulting in a density of 400 plants ha⁻¹ (Bernal, 2010).
Agronomic management
During the crop’s development and production cycle, fertilization was applied at the following annual doses: N 200 kg ha⁻¹; P 50 kg ha⁻¹; K 100 kg ha⁻¹; Ca 50 kg ha⁻¹; Mg 15 kg ha⁻¹; S 35 kg ha⁻¹; and the following micronutrients: Fe 892 g ha⁻¹; B 227 g ha⁻¹; Mn 487 g ha⁻¹; Zn 114 g ha⁻¹; and Cu 76 g ha⁻¹, along with 5 kg of compost per plant (Gaona Gonzaga et al., 2020). Fertilization was fractionated and applied every two months using solid fertilizers. Given the region's high rainfall, only weekly supplemental irrigation was provided, using a hose for individual plant watering.
Formative pruning was performed to create four productive arms, which were trained along a wire trellis. After each harvest, short pruning was applied to each arm to encourage the development of new productive shoots. Diseases, old, or vegetative branches were pruned regularly (Bernal, 2010). For insect control, Tiacloprid + Deltamethrin was applied at a concentration of 1 cm³ L⁻¹; for Botrytis cinerea control, Prochloraz was applied at 0.5 cm³ L⁻¹.
Experimental design
Grafted plants of the “Colombian” granadilla variety were used on the following rootstocks (treatments): P. edulis f. edulis (gulupa), P. edulis f. flavicarpa (yellow passion fruit), P. quadrangularis L. (badea), and P. alata Curtis. (sweet passion fruit), and with P. ligularis (granadilla) as the control.
A completely randomized design was used, with five treatments (genotypes of Passiflora from different species), and the experimental unit consisted of one plant. Each treatment included 23 replicates (plants), totaling 115 plants evaluated over an area of 2500 m² with a history of wilt caused by Fusarium sp.
Variables of evaluation
The variables rootstock diameter and scion diameter were measured using a digital caliper (Mitutoyo, Vernier 500-159-30), at 5 cm from the graft union in each corresponding case. Mortality percentage was evaluated over an 18-month period by counting the number of living and dead plants due to wilt, with the result expressed as a percentage. Yield was calculated based on the number of fruits harvested per plant over a 6-month production period. Fruit quality variables such as equatorial diameter and polar diameter were measured using a digital caliper (Mitutoyo, Vernier 500-159-30); pulp weight was determined with a digital scale (Camry, EK3252); and soluble solids content was recorded from 10 fruits per plant using a digital refractometer (Atago, PAL2), beginning at 10 months post-transplant.
Data analysis
Data were subjected to tests for homoscedasticity and normality using Levene's and Shapiro-Wilk tests, respectively. An analysis of variance (ANOVA) was then conducted to determine differences between treatments, followed by Tukey’s test at the 5% level to identify differences among means; except for the survival percentage, for which the Kruskal-Wallis test was used (Allison, 1995). All analyses were performed using the "Agricolae" package in R Studio software, version 4.4.3.
RESULTS AND DISCUSSION
F. oxysporum and F. solani are the pathogens most commonly associated with tropical fruit crops (Zakaria, 2023). The attack of Fusarium, particularly F. solani f. sp. passiflorae, on granadilla crops has been previously reported in production areas (Guerrero & Hoyos-Carvajal, 2011; ICA, 2011), and it causes low productivity in the crop (Ocampo et al., 2022). Control of this soil-borne pathogen is highly challenging, as it produces resistant spores that allow the fungus to survive chemical control measures and persist in the soil for many years (Smith, 2007). The use of resistant species as rootstocks has proven to be an effective control strategy for soil-borne pathogens or for genetic improvement in various crops (Patiño-Pacheco & Pérez-Cardona, 2021). For this reason, evaluating Passifloracea genotypes represents a viable alternative for developing a tool to curb disease attacks and reduce the need for soil-applied pesticides, thereby contributing to the prevention of contamination caused by the indiscriminate use of agrochemicals.
Table 1 shows the results for rootstock and scion diameter, compatibility index, yield, and plant survival. Regarding yield, statistical differences were observed. The rootstock with the best yield was P. edulis f. edulis, with 23.27 kg plant⁻¹ over a six-month harvest period, while the lowest yield was recorded with P. alata, with 15.88 kg plant⁻¹. In the case of the control treatment (P. ligularis), yield data could not be recorded, as the majority of the plants exhibited symptoms of wilting and therefore did not reach the productive stage. The few plants that survived produced only a small number of fruits, which were not representative enough to constitute a valid harvest.
Table 1
Rootstock and scion diameter, compatibility index, yield, and survival percentage obtained from the evaluation of the perfor-mance of four rootstocks for P. ligularis
|
Rootstocks |
Roostock diameter (mm) |
Scion diameter (mm) |
Compatibility index |
Yield (kg planta-1) |
Survival percentage (%) |
|
Passiflora edulis f. edulis(Gulupa) |
36,19 a |
32,40 a |
1,11 a |
23,27 a |
95,60 a |
|
Passiflora edulis f. flavicarpa (yellow passion fruit) |
29,52 b |
35,29 a |
0,85 b |
20,52 a |
92,00 a |
|
Passiflora quadrangularis (Badea) |
25,95 b |
35,49 a |
0,73 c |
17,48 ab |
54,17 b |
|
Passiflora alata (Sweet passion fruit) |
29,06 b |
34,09 a |
0,86 b |
15,88 b |
73,91 ab |
|
Passiflora ligularis (Granadilla - Control) |
20,80 b |
20,80 a |
------- |
------- |
8,30 c |
Grafting is a promising horticultural technology that can influence both yield and fruit quality (Habibi et al., 2022; Barik et al., 2023). In terms of yield, the two rootstocks with the highest plant survival rates (gulupa and yellow passion fruit) also had the highest yields per plant. It is worth noting that these harvest results cover a six-month period, as the primary objective of this study was to determine plant survival in the presence of wilt. Therefore, it is recommended that longer-term evaluations be conducted to determine the true productive potential of the plants, considering that granadilla is a semi-perennial species.
The greatest rootstock diameter was observed in P. edulis f. edulis (36.19 mm), while the smallest diameter was recorded in the control (20.80 mm). Regarding the scion diameter, no statistical differences were observed among the treatments, which is attributed to the fact that the same genetic material (P. ligularis) was grafted onto all four rootstocks, and there was no influence from the rootstock on the development of this parameter.
The compatibility index is an essential factor to consider when identifying a suitable rootstock. An index equal to 1 indicates compatibility, while values lower than 1 suggest incompatibility (López et al., 2008). Therefore, when the value approaches 1, the graft union is considered perfect (Vargas et al., 2018). For this parameter, P. edulis f. edulis showed the value closest to 1, although slightly higher (1.11), indicating the best compatibility with the P. ligularis scion.
The highest survival percentages were obtained with the rootstocks P. edulis f. edulis (95.60%) and P. edulis f. flavicarpa (92.00%), 18 months after transplanting (Figure 1). This contrasts sharply with the P. ligularis control plants, which exhibited a survival rate of only 8.3%. The high mortality in this treatment is believed to be associated with attacks by Fusarium sp., based on the observed symptoms of wilting and root necrosis (Figure 2).
Results showed that the two rootstocks (P. edulis f. edulis and P. edulis f. flavicarpa) with the highest plant survival rates had values close to 1 (1.11 and 0.85, respectively); however, gulupa (P. edulis f. edulis) was the genotype with the best compatibility in this study.
Londoño (2012) reported that species such as P. alata and P. quadrangularis showed no signs of damage from F. oxysporum attacks but were susceptible to F. solani. These results are consistent with the findings of this study, as the survival percentages of plants on these rootstocks were 73.91% and 54.17%, respectively. Similar trends were found by other authors, such as Pereira et al. (2019), who noted that P. alata genotypes had a higher proportion of field mortality related to Fusarium wilt. In contrast, Silva et al. (2013) found resistance in some genotypes of P. alata and P. cincinnata to Fusarium species. Moreover, Ocampo et al. (2022) suggested that the use of P. maliformis rootstocks, resistant or tolerant to F. solani, could be a viable control alternative. Pacheco-Patiño and Pérez-Cardona (2021) reported that certain geno-types of P. edulis f. flavicarpa and P. maliformis are resistant to F. solani, corroborating the results of this study, where the yellow passion fruit rootstock showed a 92.00% plant survival rate. Nonetheless, P. edulis f. edulis also achieved a high survival rate (95.60%), making it another recommended geno-type for use as a rootstock in granadilla cultivation.
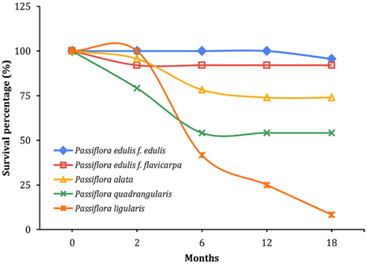
Figure 1. Survival percentage of the treatments after 18 months of evaluation.

Figure 2. Symptoms (stem necrosis and wilting) of infection caused by Fusarium sp. in a control plant of P. ligularis.
Silva et al. (2013) identified genotypes of P. edulis (both yellow and purple), P. nitida, and P. cincinnata with levels of resistance to F. oxysporum f. sp. passiflorae; while Preisigke et al. (2015) found that P. nitida, P. foetida, and P. quadrangularis exhibited the highest levels of resistance against F. solani. However, they recommended P. nitida as the best alternative due to its consistent resistance. Based on these findings, it is essential to identify the specific pathogen species in order to select the most appropriate rootstock, as resistance to Fusarium can vary within the Passiflora genus, given the highly heterozygous nature of the genotypes (Freitas et al., 2016), owing to their allogamous nature (Londoño, 2012).
Although grafting can positively influence fruit quality (Habibi et al., 2022), some studies have reported no observable effect from the rootstock (Nimbolkar et al., 2016). In this study, rootstocks did not significantly influence fruit quality traits, making them suitable for inclusion in integrated disease management strategies in commercial orchards, as they preserve the commercial variety's desired qualities.
Table 2 shows no significant differences in fruit weight or pulp weight (including seeds) among the treatments. The largest equatorial fruit diameter was recorded in P. quadrangularis (68.77 mm), while the smallest was observed in P. edulis f. edulis (65.37 mm). Equatorial diameter showed differences, which would be reflected in the fruit's shape rather than its quality. On the other hand, no significant differences were found between treatments in terms of polar diameter.
Regarding to soluble solids content, it was determined that fruits from plants grafted onto P. edulis f. edulis had the lowest value (15.75 °Brix), while no differences were observed among the other treatments. Minor differences for this parameter were observed (±0.50 °Brix approxi-mately), which would not significantly affect fruit sweetness for market purposes. The values obtained in this study fall within the 14–17 °Brix range reported in other studies (Franco Mora et al., 2008; Cañar & Caetano, 2012).
The Passifloraceae family has a high degree of natural genetic variability, a characteristic of great importance for breeding programs (Oliveira et al., 2008). Consequently, efforts should be directed at increasing the frequency of desirable resistance alleles to Fusarium through recombination among resistant plants (Patiño-Pacheco & Pérez-Cardona, 2021). Leveraging this knowledge will enable the use of these results for the development of resistant varieties or new rootstocks, aiming to reduce losses caused by wilt disease in Ecuador. In this context, it is necessary to intensify research efforts to identify reliable resistance sources and carry out interspecific hybridizations (Freitas et al., 2016) to generate new germplasm with resistance traits. However, since the development of a resistant cultivar is a slow process, existing genotypes that demonstrate resistance or tolerance should be used directly as rootstocks for the management of Fusarium-induced wilt (Pereira et al., 2019). In addition, it is important to select specific genotypes of rootstocks based on their resistance traits and overall plant performance, because they will have better response and behavior to the local disease-conditions (Antón-Domínguez et al., 2024: Naik et al., 2024).
It is relevant to research about the differential defense mechanisms between resistant and sensitive rootstocks and provide insights into plant–pathogen interactions, as well as metabolomics pathways associated with the resistance differences among the rootstock (Wang et al., 2025). All these aspects will contribute to the development of rootstooks as a strong component for promoting sustainable agriculture.
Table 2
Fruit quality variables recorded in granadilla fruits grafted onto four Passiflora genotypes
|
Rootstocks |
Fruit weight (g) |
Pulp weight (g) |
Equatorial diameter (mm) |
Polar diameter (mm) |
Soluble solids (°Brix) |
|
Passiflora edulis f. edulis (Gulupa) |
116,86 a |
71.59 a |
65,37 b |
78,10 a |
15,75 b |
|
Passiflora edulis f. flavicarpa (yellow passion fruit) |
118,53 a |
70.41 a |
66,63 ab |
78,20 a |
16,28 ab |
|
Passiflora quadrangularis (Badea) |
120,75 a |
68,50 a |
68,77 a |
78,88 a |
16,43 ab |
|
Passiflora alata (Sweet passion fruit) |
119,22 a |
70.94 a |
66,82 b |
79,89 a |
16,66 a |
CONCLUSIONS
Based on the results of this research, P. edulis f. edulis (gulupa) and P. edulis f. flavicarpa (yellow passion fruit) showed the highest survival percentages of P. ligularis plants in the field, as well as the highest yields. However, the best compatibility was observed with P. edulis f. edulis. Consequently, these two subspecies can be used as rootstocks for granadilla cultivation to mitigate the effects of plant wilting.
Moreover, fruit quality was not influenced by rootstock, indicating that the harvested fruit would be suitable for commercialization.
Finally, it is recommended to evaluate other Passiflora species such as P. maliformis, P. nitida, and P. cincinnata under the growing conditions of northern Ecuador, in order to expand the range of rootstock options for this commercially important crop.
REFERENCES
Antón-Domínguez, B. I., Arquero, O., Lovera, M., Trapero, A., Agustí-Brisach, C., & Trapero, C. (2024). Resistance of pistachio grafted on different Pistacia rootstocks against Verticillium wilt under field conditions. Plant Pathology, 73, 2466-2476. http://doi.org/10.1111/ppa.13978
Aragón. J., Albuja, M., Guzmán, j., Burbano, E., & Aragón, M. (2019). Financiamiento productivo a los agricultores en la zona de Intag, Cantón Cotacachi. Anales Científicos, 80, 355-363 (2019).
Arias, J., Ocampo, J., & Urrea, R. (2016). Sistemas de polinización en granadilla (Passiflora ligularis Juss.) como base para estudios genéticos y de conservación. Acta Agronómica, 65,197-203.
Ayres, A. S., de Araújo, L. L., Soares, T. C., Costa, G. M., Reginatto, F. H, Ramos, F. A., & Zucolotto, S. (2015). Comparative central effects of the aqueous leaf extract of two populations of Passiflora edulis. Revista Brasileña Farmacognosia, 25, 499–505. http://doi.org/10.1016/j.bjp.2015.06.007
Rodríguez, L. F., & Bermudez, L. T. (2009). Economía y gestión del sistema de producción de pasifloráceas en Colombia. In: Miranda, D., Fischer, G., Carranza, C., Magnitskiy, S., Casierra, F., Piedrahíta, W., & Flórez, L.E (eds). Cultivo, poscosecha y comercialización de las pasifloráceas en Colombia: maracuyá, granadilla, gulupa y curuba. Bogotá: Sociedad Colombiana de Ciencias Hortícolas, p. 303-326.
Barik, S., Ponnam, N., Acharya, G., Kumari, M., Adamala, A. K., Petikam, S., Sangeetha, G., Singh, T. H., Singh, H. S., & Sahu, G. S. (2023). Assessment of Bacterial wilt-resistant Solanum genetic resources as rootstocks for yield and fruit quality traits in eggplant. Australasian Plant Pathology, 52, 253–269. http://doi.org/10.1007/s13313-023-00916-w
Bernal, J. (2010). El cultivo de granadilla. Corpoica. Disponible en: https://es.slideshare.net/cristianperezperez56/cultivo-de-la-granadilla Castro, J., & Cerdas, M. (2003). Manual de producción, cosecha y postcosecha de la granadilla. San Jose, Costa Rica: Ministerio de Agricultura y Ganadería.
Cabrera, C. (2006). Caracterización de las propiedades físicas y químicas del fruto de granadilla, Passiflora ligularis Juss. Ibarra: Universidad Técnica del Norte. DANE. (2015). Encuesta Nacional Agropecuaria. Boletín técnico. Bogotá, D.C. Colombia. pdf. 25 p.
Cañar, D. Y., & Caetano, C. M. (2012). Caracterización fisicoquímica preliminar como estrategia para promoción y conservación de tres frutales neotropicales. Acta Agronómica, 61, 83–84.
Castro Vásquez, D. P. (2020). Manejo agroecológico del cultivo de granadilla (Passiflora ligularis) en el Ecuador. Tesis de Pregrado, Universidad Central del Ecuador, Quito, Ecuador, 61 p.
De Oliveira, E. J., da Silva Santos, V., Souza de Lima, D., Machado, M. D., Sales Lucena, R., Nunes Motta, T. B., & Da Silva, C. M. (2008). Seleção em progênies de maracujazeiro-amarelo com base em índices multivariados. Pesquisa Agropecuária Brasileira, 43, 1543-1549.
Di Rienzo, J., Casanoves, F., Balzarini, M., González, L., Tablada, M., & Robledo, C. (2016). InfoStat versión 2016, Grupo InfoStat, FCA, Universidad Nacional de Córdova, Argentina. Recuperado de: http://www.infostat.com.ar
Escobar, L. (1991). La sistemática y evolución de las Passifloras. En: Memorias. 1er Simposium Internacional de Passifloras. Palmira, Colombia. p. 51–54.
Espinosa, D., Melgarejo, L., Hernández, M., Melo, S., & Fernández, J. (2018). Physiological and biochemical characterization of sweet granadilla (Passiflora ligularis Juss) at different locations. Acta Horticulturae, 1194, 1459-1464.
Franco Mora, O., Tobar Reyes, J.R., Quijano Ruiz, R., & González Huerta, A. (2008). Caracterización preliminar de frutos de granada china (Passiflora ligularis Juss.) en Hueyapan y Teziutlán, Puebla. Ciencia, 15, 54-60.
Freitas, J. C. O., Viana, A. P., Santos, E. A., Paiva, C. L., Silva, F. H. L., Amaral Jr., A. T., Souza, M. M., & Dias, V. M. (2016). Resistance to Fusarium solani and characterization of hybrids from the cross between P. mucronata and P. edulis. Euphytica, 208, 493–507.
Gaona-Gonzaga, P., Vásquez-Rojas, L., Aguayo-Pacas, S., Viera-Arroyo, W., Viteri Díaz, P., Sotomayor-Correa, A., Medina-Rivera, L., Mejía-Bonilla, P., & Cartagena-Ayala, Y. (2020). Respuesta del cultivo de granadilla (Passiflora ligularis Juss) cultivar “Colombiana” al suministro de nitrógeno y potasio por fertirriego. Manglar, 17, 75-82.
Garcia, M. (2008). Manual de manejo cosecha y poscosecha de la granadilla. Bogota: Diseño, diagramación, impresión y encuadernación.
García, J., Floriano, J., & Vera, L. (2007). Enfermedades y plagas del cultivo de granadilla (Passiflora ligularis) en el departamento del Huila. Centro de Investigación Nataima, El Espinal, Tolima, CORPOICA, Colombia. p. 37.
Guerrero, E., & Hoyos-Carvajal, L. (2011). Buenas Prácticas Agrícolas (BPA) con énfasis en el manejo integrado de plagas y enfermedades de gulupa (Passiflora edulis Sims.). Bogotá, D. C. MADR, Asohofrucol, Fondo Nacional de Fomento Hortofrutícola, Universidad Nacional de Colombia, Facultad de Agronomía.
Habibi, F., Liu, T., Folta, K., & Sarkhosh, A. (2022). Physiological, biochemical, and molecular aspects of grafting in fruit trees. Horticulture Research, 9, uhac032.
Hoyos-Carvajal, L. M., & Castillo Corredor, S. Y. (2015). Enfermedades en granadilla (Passiflora ligularis Juss). In Melgarejo, L. M. (ed.), Granadilla (Passiflora ligularis juss): Caracterización ecofisiológica del cultivo. Universidad Nacional de Colombia. pp. 153-177.
Instituto Colombiano Agropecuario (ICA). Manejo de problemas fitosanitarios del cultivo de gulupa (Passiflora edulis Sims.): Medidas para la temporada invernal. Produmedios, Bogotá, Colombia, 31 p.
Joy, P. P. (2010) Passion Fruit (Passiflora edulis Sims): Passifloraceae; Kerala Agricultural University: Kerala, India; pp. 1–8.
Jorgensen, P., & Tye, A. (2017). Passifloraceae. En: León-Yánez, S., Valencia, R., Pitmam, N., Endara, L., Ulloa Ulloa, C., & Navarrete, H. (eds). Libro Rojo de Plantas Endémicas del Ecuador. Publicaciones del Herbario QCA, Pontificia Universidad Católica del Ecuador, Quito.
Khai, T. V., Thuy, V. T. B., Chau, N. T. M., & Giang, N. T. N. (2025). The impact of three rootstock types on Fusarium sp. wilt resistance in melons plants (Cucumis melo L.) in Can Tho city (Vietnam). Plant Science Today, 12, 1-7. http://doi.org/10.14719/pst.3596
Londoño, J. (2012). Evaluación de la resistencia genética de especies de Passiflora spp a Fusarium spp, agente causal de la “secadera”. Tesis Magister. Universidad Nacional de Colombia. Palmira, Colombia. 119 p.
López, M. C., Hurtado-Salazar, A., Ocampo, J., da Silva, D. F. P., & Ceballos Aguirre, N. (2023). Evaluation of purple passion fruit grafted onto a Fusarium wilt-tolerant rootstock. Pesquisa Agropecuária Brasileira, 58, e03011. http://doi.org/10.1590/S1678-3921.pab2023.v58.03011
López, J., Romo, A. R., & Domínguez, J. G. (2008). Evaluación de métodos de injerto en sandía (Citrullus lanatus (Thub.) Matsum. & Nakai) sobre diferentes patrones de calabaza. Idesia, 26, 13-18.
Martínez, E. P., Vergara, J. A., Cruz, G. N., & Aguirre, J. E. (2024). Passilora root rot: Fusarium solani f.sp. passiflorae in Colombian production systems. Temas Agrarios, 29, 225-245. http://doi.org/10.21897/q15q0254
Meletti, L., Soares, M., Bernacci, L., & Passos, I. (2005). Melhoramento genético do maracujá: passado e futuro. En Maracujá: germoplasma e melhoramento genético. Planaltina: Embrapa Cerrados. cap. 3, p. 55-78.
Molagholizadeh, F., Hajianfar, R., Saremi, H., & Moghadam, A.A. (2023). Evaluation of tomato rootstocks resistant to the fungal wilt disease caused by Fusarium oxysporum f. sp. lycopersici. Australasian Plant Pathology, 52, 195–205. http://doi.org/10.1007/s13313-023-00910-2
Naik, S. A. T. S., Hongal, S. V., Hanchinamani, C. N., Manjunath, G., Ponnam, N., Shanmukhappa, M. K., Meti, S., Khapte, P. S., & Kumar, P. (2024). Grafting bell pepper onto local genotypes of Capsicum spp. as rootstocks to alleviate bacterial wilt and root-knot nematodes under protected cultivation. Agronomy, 14, 470. http://doi.org/10.3390/agronomy14030470
Namisy, A., Chen, S. Y., Sritongkam, B., Unartngam, J., Thanarut, C., & Chung, W. H. (2025). Evaluation of luffa rootstocks to improve resistance in bitter gourd (Momordica charantia L.) against Fusarium wilt. Plants, 14, 1168. http://doi.org/10.3390/plants14081168
Navarrete, J. (2017). Estudio de la producción y comercialización de la granadilla (Passiflora ligularis) en la provincia de Imbabura (tesis pregrado). Universidad Técnica del Norte, Ibarra, Ecuador.
Nimbolkar, P. K., Awachare, C., Reddy, Y. T. N., Chander, S., & Hussain, F. (2016). Role of rootstocks in fruit production–A review. Journal of Agricultural Engineering and Food Technology, 3, 183-188.
Ocampo, J., Morillo-Coronado, Y., Espinal, F., & Moreno, I. (2022) Tecnología para el cultivo del maracuyá (Passiflora edulis f. flavicarpa Degener) en Colombia. Universidad Nacional de Colombia y Corporación Colombiana de Investigación Agropecuaria-Agrosavia Palmira. Palmira, Colombia.
Ortiz, E., & Hoyos, L. (2009). Secadera: Agentes causales y sintomatologìa asociada en gulupa (Passiflora edulis Sims.). Universidad Nacional de Colombia. 36 p.
Paredes, E. (2012). Perfil Comercial. Sierra exportadora. Arex: tu aliado para exportar. Rivera, B., Miranda, D., Ávila, L. A., & Nieto A. M. (2002). Manejo integral del cultivo de la granadilla (Passiflora ligularis Juss). Manizales: Universidad de Caldas, PRONATTA, 130 pp.
Patiño-Pacheco, M. J., & Pérez-Cardona, O. Y. (2021). Evaluación de la resistencia de genotipos de Passifloras a Fusarium solani f.sp. passiflorae en granadilla. Entramado, 17, 256-267.
Preisigke, S., Martini, F. V., Rossi, A. A. B., Serafim, M. E., Barelli, M. A. A., da Luz, P. B., Araujo, K. L., & Neves, L. G. (2015). Genetic variability of' Passiflora spp. against collar rot disease. Australian Journal of Crop Science, 9, 69-74.
Rodríguez-Carlosama, A., & Rodríguez-León. (2015). Granadilla (Passiflora ligularis Juss): Caracterización Ecofisiológica del cultivo. Departamento de Biología. Universidad Nacional de Colombia. Bogotá, Colombia.
Ríos, C. (2012). Perfil de mercado de la granadilla (Passilfora ligularis) (tesis de pregrado) Universidad Nacional José Faustino Sánchez Carrión, Perú.
Sierra-Escobar, J. A., Navarro Alzate, R. A., & Yepes, G. J. (2017). Efectos de la interacción de hongos micorrizo arbusculares (HMA) y Meloidogyne javanica en plántulas de granadilla (Passiflora ligularis Juss). Bionatura, 2, 21-26.
Silva, A. D. S., Oliveira, E. J. D., Haddad, F., Laranjeira, F. F., Jesus, O. N. D., Oliveira, S. A. S. D., de Carvalho Costa, M. A. P., & Freitas, J. P. X. D. (2013). Identification of passion fruit genotypes resistant to Fusarium oxysporum f. sp. Passiflorae. Tropical Plant Pathology, 38, 236-242.
Smith, S. N. (2007). An overview of ecological and habitat aspects in the genus Fusarium with special emphasis on the soil-borne pathogenic forms. Plant Pathology Bulletin, 16, 97–120.
Tamayo. P., Giraldo, B., & Morales, J. (1999). Enfermedades en semilleros y almácigos de granadilla. Revista Facultad Nacional de Agronomía Medellín, 52, 773- 779,
Vargas, Y., Nicolalde, J., Alcívar, W., Moncayo, L., Caicedo, C., Pico, J., Ron, L., & Viera, W. (2018). Response of wild solanaceae to Meloidogyne incognita inoculation and its graft compatibility with tree tomato (Solanum betaceum). Nematrópica, 48(2), 126-135.
Varón, E., Santos, O., Vera, Á., & Salamanca, J. (2009). Problemas fitosanitarios de secadera y mosca de los botones florales en el cultivo de la granadilla. Colombia. Corpoica. 52 p.
Viera, W., Shinohara, T., Samaniego, I., Terada, N., Sanada, A., Ron, L., & Koshio, K. (2022a). Pulp mineral content of passion fruit germplasm grown in Ecuador and its relationship with fruit quality traits. Plants, 11, 697. http://doi.org/10.3390/plants11050697
Viera, W., Shinohara, T., Samaniego, I., Sanada, A., Terada, N., Ron, L., Suárez-Tapia, A., & Koshio, K. (2022b). Phytochemical composition and antioxidant activity of Passiflora spp. germplasm grown in Ecuador. Plants, 11, 328. http://doi.org/10.3390/plants11030328
Viera, W., Shinohara, T., Sanada, A., Terada N., & Koshio, K. (2025). Physical and chemical characterization of passion fruit, focusing on the differences in juice carotenoids and sugars. The Horticulture Journal, 94, 190-199. http://doi.org/10.2503/hortj.QH-150
Wang, L., Yi, Q., Yu, P., Kumar, S., Zhang, X., Wu, C., Weng, Z., Xing, M., Huo, K., Chen, Y., & Guopeng Zhu. (2025). Rootstock selection for resisting cucumber Fusarium wilt in Hainan and corresponding transcriptome and metabolome analysis. Plants, 14, 359. http://doi.org/10.3390/plants14030359
Yockteng, R., Coppens, G., & Souza, T. (2011). Passiflora L. Springer. Verlag, Berlin. 129 pp.
Zakaria, L. (2023). Fusarium species associated with diseases of major tropical fruit crops. Horticulturae, 9, 322. http://doi.org/10.3390/horticulturae9030322